Abstract
BACKGROUND
Bone marrow examination is essential in diagnosis, staging and monitoring of various hematologic disorders. The aspirate smears and core biopsy are complementary samples; current clinical benchmarks recommend an optimal core sample length of at least 15-20 mm. We assessed the core length in 2 cross-sectional cohorts from 2001 and 2011 in 32 academic medical centers from the US and Canada, the first such study to date.
METHODS
After IRB approval, participants collected data from pathology reports (including the preprocessing length) and measured aggregate postprocessing and evaluable marrow length using a uniform validated methodology on 100 consecutive marrow samples in 2001 and 2011 at each institution. Deidentified data was centralized at Roswell Park Cancer Institute (RPCI) and centers were anonymized. A total of 6374 samples were accepted for statistical analysis, performed using SAS (v. 9.4 or higher; SAS Institute, Cary, NC) at RPCI. Relationship between core length and NCCN status, geographic location, gender, age, and staging was assessed using the PROC MIXED and PROC GLIMMIX procedure using a random center effect and a nominal significance level of 0.05.
RESULTS
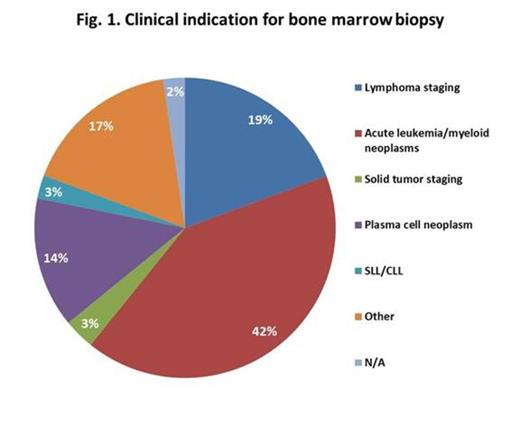
The study cohort included 56% men and 44% women, mean age 51 (range, 1-102) years, 88% adults (³18) and 12% children (<18). The two most common clinical indications were acute leukemia/myeloid neoplasms and lymphoma staging (Fig 1).
A core biopsy was obtained in 90% of 2001 and in 95% of 2011 samples. Most cores were unilateral (85%); bilateral sampling decreased from 10% to 4% between 2001 and 2011. An aspirate specimen was received in the pathology department in 86% of the cases; however, a clot section was prepared in only 56% of the cases.
Preprocessing core length (PreCL; n=3141) documentation in pathology reports was missing in 9 centers for both years; in 3 centers it was available only for one year; its mean (standard deviation) was 16.9 (9.9) mm, decreasing from 2001 to 2011 [17.7 (11.9) to 16.2 (8); p=0.002].
Postprocessing core length (PostCL; n=5742) mean (SD) was 14.2 (8.5) mm, significantly shorter in women than in men (p<0.001), longer in bilateral than unilateral (p<0.001) and in staging than in non-staging samples (p<0.001), in children than adults (NS), and in 2001 than 2011 samples [14.3 (9.4) v. 14.2 (7.6); p=0.144].
Evaluable marrow space length (EML; n=5617) mean (SD) was 10.7 (7.5) mm and decreased from 2001 to 2011 [10.9 (8.5) to 10.6 (6.6); p=0.002]. Ninety-one samples (1.6%) with measureable PostCL were entirely devoid of evaluable marrow.
PostCL was 15% shorter than PreCL. The EML measured 24% less than the PostCL and 33% less than the PreCL.
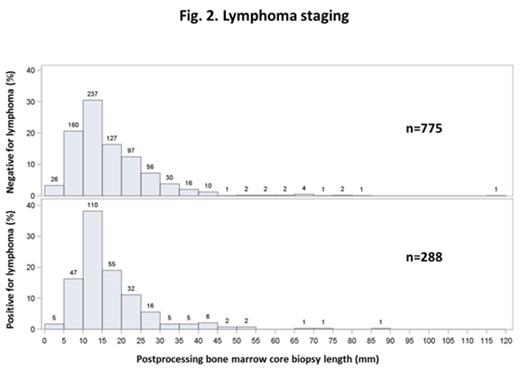
Lymphoma staging samples (n=1222) were obtained from adults (96%) and children (4%); of these, only 45% and 28% reached the PostCL of 15 mm and 20 mm, respectively. Lymphoma involvement rate was 16%, 25%, 32%, 30%, and 23% when PostCL was <5, 5-10, 11-14, 15-20, and ³20 mm, respectively (Fig 2).
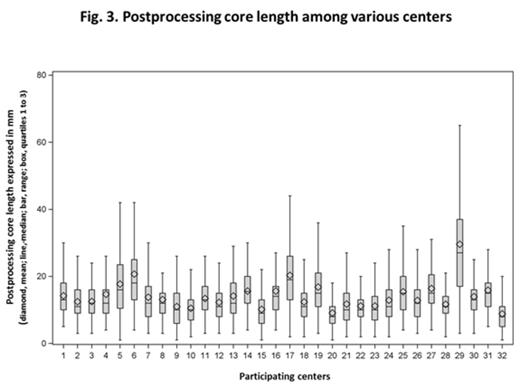
There was no significant difference of PostCL mean based on geographic region or NCCN status. PostCL mean ranged from 8.8 to 29.6 mm and its median was ³15 mm in only 12 of 23, 9 of 32, and 2 of 32 centers for PreCL, PostCL, and EML, respectively (Fig 3).
When compared to current benchmarks, only 53%, 38%, and 22% of all samples were ³15 mm and only 30%, 19%, and 10% were ³20 mm for PreCL, PostCL, and EML, respectively.
CONCLUSIONS
Current benchmarks for bone marrow core biopsy adequacy are met in only a minority of cases. Samples from women were shorter than from men. Bilateral sampling decreased and PreCL and EML diminished significantly between 2001 and 2011, while PostCL showed only minimal decrease. This discrepancy may be due to technical differences. Although staging cores were longer than non-staging ones, most did not meet the benchmarks. The wide variability among different centers suggests significant institutional differences and lack of standardization in bone marrow sampling.
Over a third of all centers did not record preprocessing length, an important parameter which may guide operator assessment of adequacy at the bedside. PostCL and/or EML can be measured by the pathologist and integrated in the report to document adequacy. In staging samples, a minimal marrow space sampling should be required to avoid understaging. Consensus definition of an inadequate core biopsy is currently lacking but would be desirable for uniform pathology reporting, clinical protocols and management.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal