Abstract
Background: AITL is a common subtype of peripheral T-cell lymphoma (PTCL) that typically presents with lymphadenopathy and extranodal disease and is associated with frequent infections due to immune dysregulation. Patients with AITL generally have a poor prognosis, even with aggressive chemotherapy. Romidepsin is a structurally unique, potent, bicyclic, class 1 selective histone deacetylase inhibitor approved by the US Food and Drug Administration for the treatment of patients with PTCL who have received at least 1 prior therapy and patients with cutaneous T-cell lymphoma who have received at least 1 prior systemic therapy. A phase 2, single-arm, open-label registration study (GPI-06-0002) demonstrated durable clinical benefit and long-term tolerability of romidepsin in patients with relapsed or refractory PTCL. Here, we present updated data for patients with AITL from GPI-06-0002.
Methods: Patients with histologically confirmed PTCL (N = 130) who experienced failure with or were refractory to ≥ 1 prior systemic therapy received romidepsin 14 mg/m2 as a 4-hour intravenous infusion on days 1, 8, and 15 every 28 days for up to 6 cycles; treatment could be extended for patients with stable disease (SD) or response. The protocol was amended to allow for (but not mandate) maintenance dosing of twice per cycle for patients treated for ≥ 12 cycles; dosing could be further reduced to once per cycle at ≥ 24 cycles in patients who had received maintenance dosing for ≥ 6 months. The primary endpoint was the rate of confirmed/unconfirmed complete response (CR/CRu) as determined by an independent review committee based on International Working Group criteria. Secondary endpoints included objective response rate (ORR: CR/CRu + partial response [PR]), duration of response (DOR), and time to progression; progression-free survival and overall survival (OS) were also assessed. Disease response was assessed every 2 treatment cycles. The analysis herein is focused on updated data (median follow-up, 22.3 months) in patients with AITL.
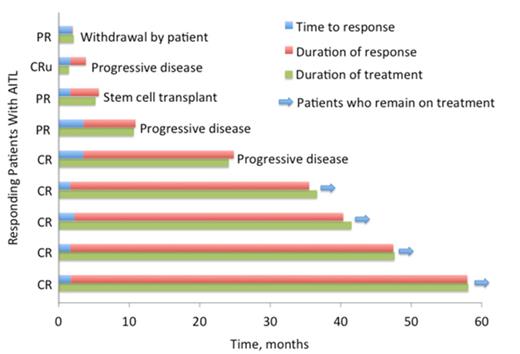
Results: Of 27 patients with AITL, most had advanced disease (96% stage III/IV; 44% with bone marrow involvement; 52% with elevated lactate dehydrogenase) and heavy pretreatment (median, 2 [range, 1-8] prior therapies) and 37% were refractory to their last line of therapy. The ORR for patients with AITL was 33% (9 of 27 patients), with most responders achieving CR/CRu (6 of 27 patients; 22%). Most responses were noted at the first response assessment, with a median time to response of 52 days. Furthermore, an additional 8 patients with AITL achieved SD (30%), 3 of whom had disease stabilization for ≥ 90 days. The median DOR has not been reached, with the longest response ongoing at 56 months (Figure). Five patients with AITL and DOR of ≥ 12 months with romidepsin had either 1 (n = 2) or 2 (n = 3) prior therapies, and 3 of the 5 were refractory to their last line of therapy (CHOP [cyclophosphamide, doxorubicin, vincristine, prednisone], GVD [gemcitabine, vinorelbine, doxorubicin], or pralatrexate). All 5 responding patients who remained on treatment for ≥ 12 cycles received maintenance dosing of twice per cycle. Dosing for the 3 patients with the most durable responses was also later amended to 1 dose per cycle. For all patients with AITL treated with romidepsin, the median OS was 18 months (range, 2-58 months). Grade 3/4 infections (all types pooled, regardless of relationship to study drug) were reported in 6 patients (22%), and no discontinuations due to infection occurred.
Conclusions: Single-agent romidepsin induced rapid, complete, and durable responses in some patients with relapsed/refractory AITL, with several responses ongoing for > 3 years. Patients with long-term responses to romidepsin received maintenance dosing. These results support the use of romidepsin in relapsed/refractory AITL.
Patients With AITL Who Achieved a Response to Romidepsin
Pro:Celgene: Honoraria. Horwitz:Bristol Myers Squibb,: Consultancy; Amgen: Consultancy; Spectrum: Research Funding; Seattle Genetics: Consultancy, Research Funding; Kiowa Kirin: Research Funding; Infinity: Research Funding; Millennium: Consultancy, Research Funding; Celgene: Consultancy, Research Funding; Jannsen: Consultancy. Prince:Celgene: Honoraria, Research Funding. Foss:Celgene: Consultancy, Research Funding, Speakers Bureau. Sokol:Celgene: Consultancy. Morschhauser:Spectrum: Honoraria; Bayer: Honoraria; Mundipharma: Honoraria; Genentech: Honoraria; Gilead: Honoraria. Pinter-Brown:Celgene: Consultancy. Padmanabhan Iyer:Janssen Biotech, Inc.: Honoraria; Celgene: Speakers Bureau; Houston Methodist Cancer Center: Employment. Shustov:Celgene: Consultancy, Honoraria, Research Funding. Balser:Celgene: Consultancy. Coiffier:Celgene: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal