Abstract
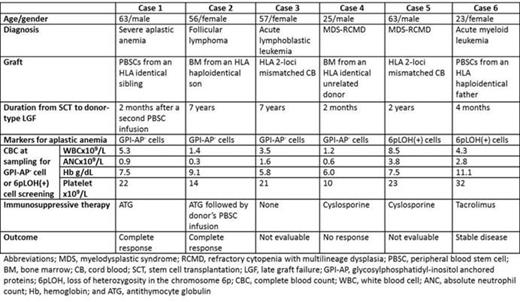
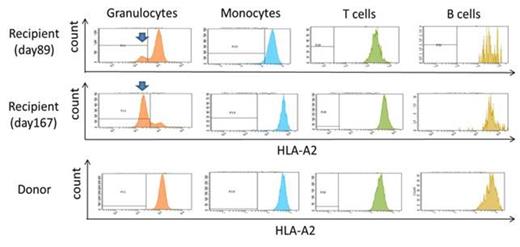
Late graft failure (LGF) without evidence of residual recipient cells is a serious complication after allogeneic hematopoietic stem cell transplantation (allo-SCT). Various factors, such as infections and chronic graft-versus-host disease (GVHD), are involved in the development of such donor-type LGF, and it is therefore difficult to identify the exact cause and to restore the graft function without performing a second transplant. One exceptionally treatable condition is aplastic anemia (AA), which occurs during donor-derived hematopoiesis and can be diagnosed by the identification of disease-specific markers in the peripheral blood. These include glycosylphosphatidyl inositol-anchored protein (GPI-AP)-deficient blood cells (Mochizuki, el al. Blood, 2008) and HLA-allele lacking leukocytes (HLA-LLs) due to copy-number neutral loss of heterozygosity in the short arm of chromosome 6 (6pLOH, Katagiri, et al. Blood, 2011) or missense mutations in the HLA alleles, which can be found when the pancytopenia develops. During the six-year period from 2007 through 2013, 12 patients with LGF after allo-SCT were referred to our clinic for the diagnosis of the pathogenesis of LGF. Three of the 12 patients were diagnosed to have late graft rejection due to the residual lymphocytes of recipient origin. None of these patients and other three patients with complete chimerism showed either GPI-AP- cells or HLA-LLs. On the other hand, either of the markers were detected in the remaining six patients with complete donor chimerims who developed LGF two to 84 months after allo-SCT after they had achieved complete hematological recovery with a platelet count > 100 x109/L. The original diagnoses included severe AA in one patient and hematological malignancies in the other five patients (Table). A range of 0.016% to 0.2% GPI-AP- granulocytes were detected in four patients, while 6.5%-80% HLA-LLs were detected in two patients (Figure). All patients first developed thrombocytopenia and gradually progressed to pancytopenia. The donors of the three patients possessed one of the four HLA class I alleles (A*02:01 and A*02:06) that we previously identified as genes associated with a susceptibility to AA. The HLA-A allele (A*02:01) that was lost in the donor's granulocytes in Case 6 was not shared by the recipient. ATG was given to two patients (cases 1 and 2) and induced complete resolution of the LGF. The thrombocytopenia of Case 6 stopped progressing at three months after allo-SCT following a dose-escalation of tacrolimus and became stable thereafter. These findings suggest that donor-type AA is a common cause of LGF with complete donor chimerism after allo-SCT. This complication occurs regardless of the recipient's original disease, and is potentially curable by immunosuppressive therapy without the need for a second SCT. It is therefore important to screen the peripheral blood for the presence of GPI-AP- cells and HLA-LLs when donor-type LGF or thrombocytopenia occurs after a patient has achieved complete hematological recovery. Possessing HLA class I alleles associated with an AA susceptibility by donors may confer a risk of developing AA to recipients.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal