Abstract
Allogeneic stem cell transplantation (AlloSCT) from HLA-matched unaffected sibling donors (MSD) has been successful for high-risk SCD, and is the only known curative therapy (Freed/Cairo et al, BMT, 2012). We have recently demonstrated 100% event free survival and absence of sickle cell symptoms following reduced toxicity conditioning and HLA matched sibling bone marrow or cord blood AlloSCT (Bhatia/Cairo et al, BMT, 2014). However, 5 out of 6 children who might benefit from this therapy lack an HLA matched family donor. Identifiable matched unrelated adult donors (URD) in this ethnic group are extremely limited and results from unrelated cord blood transplants are poor (Radhakrishnan K/Cairo et al., BBMT 2013, Kamani et al., BBMT, 2012). We previously demonstrated the use of positive CD34 selection followed by T cell add back (2 x 105 CD3/kg) from unrelated donors in pediatric recipients with both malignant and nonmalignant disease lead to 100% engraftment with minimal acute GVHD (aGVHD). In a high-risk FHI TCD thalassemia study, 16/22 cases engrafted without aGVHD and with 90% overall survival (Sodani et al., Blood, 2010). FHI TCD AlloSCT could expand the donor pool and improve outcomes for patients with high risk SCD.
This SCD consortium trial is investigating the safety, feasibility, EFS, donor chimerism, graft failure, aGVHD and chronic GVHD (cGVHD), and infectious mortality after FHI TCD AlloSCT in high-risk SCD patients (Figure 1).
High risk features included one or more of the following: ≥1 CVA, ≥2 ACS, ≥3 VOC in past 2 years, or 2 abnormal TCDs. Patients (2-20.99 yrs) without an 8/8 HLA MSD or URD and who have ≥1 high-risk SCD features were eligible. Patients received hydroxyurea 60 mg/kg/d and azathioprine 3mg/kg/d, day -59 – day -11, fludarabine (30mg/m2/d x5d), busulfan (3.2 mg/kg/d x4d [<4yrs of age: 4 mg/kg/d x4d]), thiotepa (10 mg/kg/d x1d), cyclophosphamide (50mg/kg/d x4d), R-ATG (2mg/kg/d x4d), and TLI (500cGy) followed by FHI T-cell depleted AlloSCT. AGVHD prophylaxis included tacrolimus single agent. We utilized the CliniMACS (IND 14359) to enrich for peripheral blood hematopoietic progenitor cells (HPC's); target dose of 10 x 106 CD34+ cells/kg with 2 x 105 CD3+ T cells/kg added back as a final CD3/kg concentration.
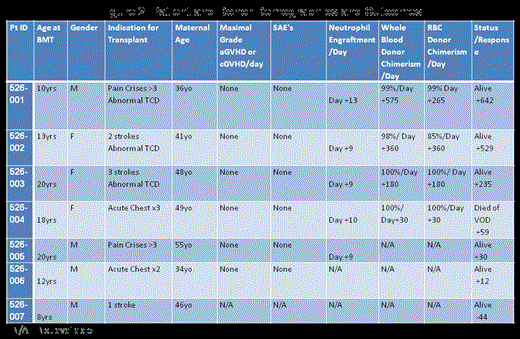
Six patients have received AlloSCT to date (Figure 2). All patients utilized maternal donors who encountered no complications during collection. All had early neutrophil engraftment (median day +9), ≥98% whole blood chimerism and ≥85% RBC donor chimerism, no aGVHD or cGVHD (Figure 2). One patient developed late hepatic SOS and died at day +59; the remainder are alive and free of disease (day +12 to +642). One more patient has been enrolled and has begun conditioning and others are in the early part of their transplant process.
Early results indicate FHI TCD AlloSCT is feasible in high-risk SCD patients who lack a MSD or URD. A larger cohort with longer term follow-up is needed to assess long-term safety and outcomes (Supported by FDA 5R01FD004090 and a grant from Otsuka) (IND #14359 and NCT 01461837). http://www.sicklecelltransplantconsortium.org
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal