Abstract
Background: We investigated the potency, biological mechanisms of action, and described the role of MYC in cell death to the proteasome inhibitor, ixazomib (MLN2238), in TCL and HL in vitro and in vivo tumor models. In addition, we sought to identify molecular biomarkers that were associated with response/resistance to ixazomib.
Methods: TCL cell lines (Jurkat, Hut78, HH) and HL cell lines (L428, L540, L1236) were treated with ixazomib for 24-72 hours, cell viability and apoptosis were analyzed by MTT and Annexin/PI by flow cytometry (FC). In vivo anti-tumor activity and survival of tumor bearing SCID mice were determined using xenografts derived from Jurkat (TCL) and L540 (HL) cell lines. Gene expression profiling (GEP) was performed using Human Affymetrix 2.0 HT array, which included Gene Set Enrichment Analysis (GSEA), for Jurkat, L540, and L428 cell lines.
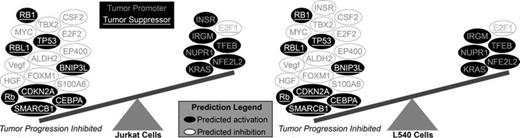
Results: Treatment with 20-100 nanomolar (nM) of ixazomib resulted in time- and dose-dependent cytotoxicity and apoptosis in all TCL and HL cell lines with all IC50’s <75nM. In vivo experiments with SCID tumor xenografts showed significant inhibition of tumor growth (P<0.001) with significantly improved survival (P<0.001) in both Jurkat and L540 models with ixazomib-treated mice versus controls. GSEA of GEP in ixazomib-treated Jurkat (TCL) and L540 (HL) cells showed significant overlap in biological functions involved in regulation of cell cycle, chromatin modification, and DNA repair processes; however lack of such conserved overlap was observed in the relatively ixazomib-resistant cell line, L428. Furthermore, the predicted activation and inhibition status of tumor suppressors and oncogenes based on GEP results showed that the balance between these processes strongly favored ixazomib treatment and overall inhibition of tumor progression (see Figure 1 ). Most notably, ixazomib prominently down-regulated protein levels of MYC and its target genes in both Jurkat (TCL) and L540 (HL) cells. Conversely, there was an increase in MYC protein levels without changes in its target gene expression in resistant L428 cells. Additionally, experiments showed that histone acetylation and lysosomal functions affected MYC levels and cell death response to ixazomib. Furthermore, inhibition of HDAC with belinostat (at 250nM) resulted in synergistic cell death with ixazomib treatment (combination index=0.8) in L428 as determined using Calcusyn software. Finally, GEP data from ixazomib-treated cell lines Jurkat, L540 and L428 using Ingenuity pathway analysis, identified CD68, TNFS10, and PSAP as biomarkers that were for ixazomib response/resistance. Further experiments with L428 (HL) cells with acquired resistance to ixazomib validated these genes as critical biomarkers of ixazomib sensitivity.
Conclusions: Ixazomib induced potent cell death at nanomolar and concentrations in TCL and HL cell lines and showed antitumor activity in vivo xenograft SCID models. Results with ixazomib from GEP and GSEA studies showed global changes in gene expression consistent with inhibition of tumor progression in both TCL and HL cells. While ixazomib treatment down-regulated MYC and its downstream substrates in TCL and HL cells, resistance to ixazomib treatment appeared to occur through a MYC-dependent mechanism. In addition, we identified several potential novel biomarkers of ixazomib response/resistance in TCL and HL. Continued examination of ixazomib in lymphoma is warranted and rational combinations with MYC and HDAC inhibitors should be explored.
GSEA and upstream regulation analysis based on differentially expressed genes following ixazomib treatment predict dominant activation of tumor suppressors and inhibition of tumor promoters, in favor of strong inhibition of tumor progression in Jurkat (TCL) and L540 (HL).
GSEA and upstream regulation analysis based on differentially expressed genes following ixazomib treatment predict dominant activation of tumor suppressors and inhibition of tumor promoters, in favor of strong inhibition of tumor progression in Jurkat (TCL) and L540 (HL).
Evens:Takeda Pharmaceutical Company Ltd: Consultancy, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal