Abstract
Minimal residual disease (MRD), as assessed by flow cytometry is a powerful predictor of outcome in multiple myeloma (MM). We and others have previously demonstrated that such analyses are informative in patients treated with autologous stem cell transplant (ASCT) and non-transplant regimens. It predicts outcome in patients in conventional CR and is applicable to patients with standard and adverse risk cytogenetics. As a consequence MRD assessment is under consideration as a surrogate endpoint for clinical trials. This is urgently needed in MM as >5yrs follow-up is typically required to demonstrate survival differences in trials of upfront therapy. If surrogate end points are to be used in clinical trials it is essential that a reproducible effect is demonstrable using multivariate models. Previous studies have confirmed the effect of MRD on PFS but a consistent effect on OS has been not been definitively shown. This may in part be due to the availability of effective salvage therapy but it is also possible that the traditional threshold of 10-4 for analysis and the categorization of patients as MRD-postive or negative is suboptimal. Flow cytometry does provide a quantitative assessment of residual tumour over a large range and the degree of tumour depletion may be more informative than a positive-negative analysis.
397 patients from the MRC Myeloma IX trial were included in this analysis. Patients were randomly assigned to CTD (cyclophosphamide, thalidomide, and dexamethasone) or CVAD (cyclophosphamide, vincristine, doxorubicin, and dexamethasone) induction for 4-6 cycles followed by standard high-dose melphalan (HDM) ASCT. BM aspirates were obtained at day 100 for MRD analysis. 500,000 cells were evaluated with six-colour antibody combinations including CD138/CD38/CD45/CD19 with CD56/CD27 in all cases and CD81/CD117 in additional cases as required. PFS and OS data analysis was landmarked from the date of the MRD assesment.
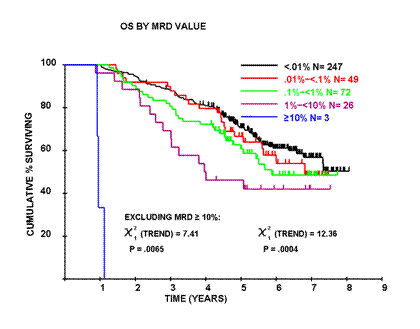
Of the 397 patients with MRD data available at day 100 after ASCT, 247/397 (62.2%) achieved <0.01% MRD. The level of residual disease varied across four logs in MRD-positive patients (0.01-<0.1% in 49/397, 0.1-<1% in 72/397, 1-<10% in 26/397 and ≥10% in 3/397). The PFS and OS for individuals with ≥1% residual disease was comparable to individuals with a PR/MR/SD confirming that MRD assessment is most relevant in CR. The level of MRD correlated with outcome. The median PFS for patients with ≥10% MRD at day 100 after ASCT was 0.8 years, with 1-<10% MRD was 1.7 years, with 0.1-<1% MRD was 1.9 years, with 0.01-<0.1% MRD was 2.7 years and for patients with <0.01% MRD was 3.1 years (P<0.001). The median OS for these groups was 1 yr, 4 yrs, 5.9 yrs, 6.8 yrs and for patients with <0.01% MRD not reached with >7.5 yrs median follow-up (P<0.001, see figure).
A Cox proportional hazards model was used to further evaluate factors influencing outcome. B2M and MRD were log-transformed and along with age were considered as continuous variables. ISS, haemoglobin (<115g/l), platelets (<150x10^9/l) and cytogenetics were used as stratification factors. Cytogenetic groups were classified as unfavourable for patients with gain(1q), del(1p32), t(4;14), t(14;20), t(14;16), and del(17p), or favourable for hyperdiploidy, t(11;14) and t(6;14), or unknown/inevaluable. MRD assessment (χ2 11.8, P=0.0006) and cytogenetics (χ2 35.5, P=<0.0001) were the only factors that retained significance in this multivariate model. Conventional categorical response, ISS and B2M were not predictive of OS (p=0.99, 0.16 and 0.56 respectively).
We would conclude that MRD quantitation is more informative than a positive or negative categorization with a 10-4 threshold and independently predicts outcome. In this analysis we were able to demonstrate an approximate 1 year survival benefit per log tumour depletion. A lower cutpoint for predicting improved outcome was not reached and more sensitive assays will likely improve outcome prediction further. This data strongly supports the role of MRD assessment as a surrogate endpoint in clinical trials.
Rawstron:Celgene: Consultancy; BD Biosciences: Consultancy, Intrasure Patents & Royalties. Gregory:Celgene: Consultancy. Davies:Celgene: Consultancy, Honoraria; Janssen-Cilag: Consultancy, Honoraria; Novartis: Consultancy. Cook:Celgene: Consultancy, Honoraria, Research Funding; Janssen-Cilag: Consultancy, Honoraria. Jackson:Celgene: Honoraria; Janssen-Cilag: Honoraria. Morgan:Celgene: Consultancy, Honoraria, Research Funding; Janssen-Cilag: Consultancy, Honoraria; Merck: Consultancy, Honoraria; Novartis: Consultancy, Honoraria. Owen:Celgene: Consultancy, Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal