Abstract
BACKGROUND: Primary Mediastinal large B-cell lymphoma (PMBL) is a rare form of Non Hodgkin Lymphoma (NHL) representing 2% of mature B-cell non-Hodgkin lymphoma in patients less than 18 years of age (Lones/Cairo et al, JCO 2000; Burkhardt et al, BJH 2005). PMBL has histological features somewhere between Diffuse Large B-Cell Lymphoma (DLBCL) and classical HL (cHL) (Abramson et al, Blood 2005). Gene expression studies suggested that the molecular signature of PMBL had a striking resemblance to the expression profile of cHL (Rosenwald et al, JEM 2003). We have recently reported that a significant decrease in EFS among children and adolescent PMBL patients compared with other stage III non-PMBL pediatric DLBCL patients following FAB/LMB 96 therapy, suggesting that children and adolescent with PMBL required alternative treatment strategies (Gerrard/Cairo et al, Blood 2013). PMBL has been demonstrated to have an over-activated NF-kB pathway by gene expression profiling (Rosenwald et al, JEM 2003). Since over 95% of PMBL express CD20, targeting the CD20 receptor with a CD20 antibody is of high clinical interest. Obinutuzumab (GA101) is novel glycoengineered anti-CD20 targeted monoclonal antibody recognizing a unique CD20 type II epitope and it has been demonstrated to have greater efficacy in reducing tumor size, inducing remission and improving survival in other B-NHL xenograft models (Mössner et al, Blood 2010). Obinutuzumab has been recently approved by FDA for first line treatment of chronic lymphocytic leukemia (CLL) in combination with chlorambucil.
OBJECTIVES: We hypothesize that obinutuzumab may be a future potential targeted agent for the treatment of PMBL, and therefore, we investigated whether obinutuzumab treatment results in significant changes in signaling pathways, genes expression, programmed cell death and cell proliferation in PMBL.
METHODS: Karpas-1106P cells (DSMZ) were treated with obinutuzumab (generously provided by Dr. Klein, Roche) at every 24 hours (1-100ug/ml). qRT-PCR, western blot, MTS, Caspase 3/7 assay (Promega) and FACS analysis were performed. The BeadChip array (Illumina, HumanHT-12) was used for gene expression profiling.
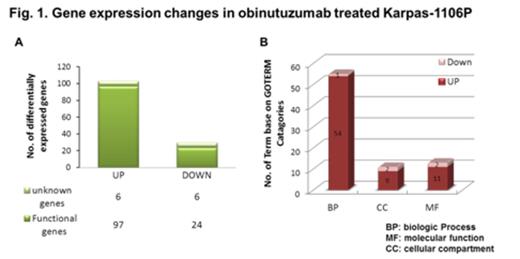
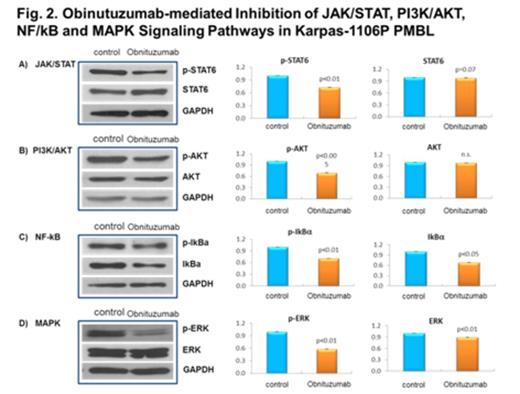
RESULTS: There was a significant decrease of cell proliferation in obinutuzumab-treated Karpas cells with 10ug/ml (0.69 ± 0.025, p<0.005) vs control (1.00 ± 0.000) at 48 hours. Concomitantly, there was a significant increase in programmed cell death in 10ug/ml obinutuzumab treated Karpas (37.80 ± 10.096, p<0.05) vs control (1.19 ± 0.762) at 48 hours. We also observed a significant decrease of CD20 expression (0.74± 0.010, p<0.05) with 10ug/ml obinutuzumab treatment at 48 hours. A total of 133 differentially expressed genes were identified by gene expression profiling (>1.5-fold, 0.57%) and 77.5% of genes including apoptosis related genes (CASP2 and PAK2) and MAPK signaling pathways (RASA1 and JUN) and EGR1 were upregulated and 22.5% of genes including ID3, GRAP and RAB6B were downregulated in obinutuzumab treated Karpas vs control (Fig 1). There were significant decreases of p-STAT6 (0.72± 0.011, p=0.01), p-Akt (0.69± 0.011, p<0.05), p-ikBα (0.70± 0.017, p<0.05) and p-Erk (0.56± 0.019, p<0.05) with 10ug/ml obinutuzumab treatment at 48 hours (Fig 2). Additionally, There were significant down-regulation of mRNA expression of Bcl-xL (0.91±0.011, p<0.04) and Bax (0.66±0.022, p<0.02) vs control.
CONCLUSIONS: We observed that obinutuzumab significantly inhibited cell proliferation and induced programmed cell death and downregulated downstream of PI3k/Akt and NF-kB signaling pathways. Gene expression analysis indicated obinutuzumab induced changes in the expression of genes in Karpas that were involved in apoptosis and signaling pathways including CASP2, EGR1 and ID3. Future studies 1) will investigate the efficacy of combination therapies to enhance programmed cell death, and 2) will assess the proteomic signature induced by obinutuzumab in obinutuzumab sensitive and resistant PMBL, and furthermore, 3) will focus on the in vivo effects of obinutuzumab in a NOD/SCID PMBL xenograft mouse model. Obinutuzumab may be a future potential targeted agent for the adjuvant treatment of PMBL lymphoma.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal