Abstract
The iNKT cell (invariant Natural Killer T cell) is a unique immune cell that has been shown to be associated with chronic inflammation in sickle cell disease. One approach to reducing iNKT mediated inflammation would be to reduce the number of iNKT cells in the tissue of SCD patients. NKTT120 is a humanized monoclonal antibody directed to the unique T cell receptor of invariant NKT cells that can deplete iNKT cells by antibody dependent cellular cytotoxicity. It recently completed a first in man Phase 1 study that demonstrated that intravenous single doses in adults with SCD specifically reduced iNKT cells without dose limiting toxicity in the therapeutic range of 0.3 and 1.0 mg/kg.
Although intravenous dosing was found to be effective and safe, the ability to dose patients by subcutaneous administration would be an asset to a chronic treatment strategy and patient compliance. The purpose of this study was to assess the bioavailability and the efficacy of NKTT120 administered subcutaneously (SC) to cynomolgus monkeys. Female adult cynomolgus monkeys were administered NKTT120 as a single subcutaneous dose of 0.03, 0.1, 0.3 mg/kg (n=3 each) or as a single intravenous (IV) dose of 0.3 mg/kg (n=3). Animals were monitored for iNKT cell depletion of peripheral blood by FACS analysis and NKTT120 concentration was monitored by ELISA.
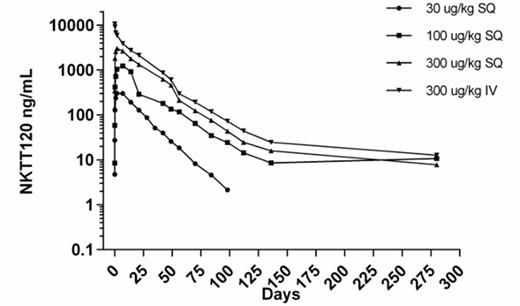
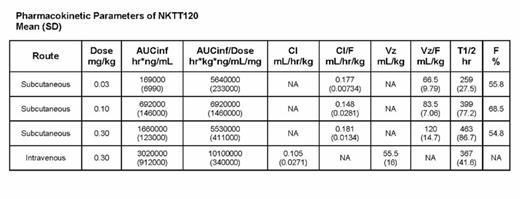
All doses resulted in complete depletion of blood iNKT cells without any change in other blood cells. The return of measureable peripheral blood iNKT cells occurred in each group when the plasma concentration was ≤ 100 ng/mL. The time to recovery was dose dependent with measureable iNKT cells found at 4-6 weeks with the 0.03 mg/kg dose and 8-14 weeks with the 0.3 mg/kg IV and SC doses. NKTT120 was dose proportional with time to maximum plasma concentration of 48 hours following SC dosing. Bioavailability of NKTT120 after SC dosing was ~60% and T1/2 was 15 days for the IV dose group and ranged from 11-19 days for the SC dose groups. The duration of iNKT cell depletion and time to recovery for the 0.3 mg/kg SC dose was similar to that observed for the 0.3 mg/kg IV dose group.
The results support a SC dosing strategy for NKTT120 and suggest that similar pharmacodynamic responses could be expected when a therapeutic intravenous dose (0.3 mg/kg) is administered by the subcutaneous route in SCD patients every 3-4 months.
Plasma Concentration vs. Time for NKTT120 Following IV or SC doses to Cynomolgus Monkeys
Schaub:NKT Therapeutics: Employment, Equity Ownership. Thariath:NKT Therapeutics: Employment, Equity Ownership. Scheuplein:NKT Therapeutics: Equity Ownership. Mashal:NKT Therapeutics: Employment, Equity Ownership.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal