Abstract
Tumour immune surveillance requires both the recognition of malignant cells as immunogenic and the functional cytotoxic capacity of an effector cell. Key cytotoxic effector cells are T lymphocytes, capable of mediating tumour cell death through granule exocytosis and death receptor signalling. Whether chemotherapy can modify immune surveillance in haematological malignancies is not yet well understood. Remission induction chemotherapy induces rapid cytoreduction of leukaemia and has the modifying potential to affect effector-target ratios and tumour cell recognition. Professional antigen presenting cells may take up the necrotic and apoptotic tumour cells, process tumour antigens, and present these to T lymphocytes alongside the costimulatory molecules required for effective priming. Thus, the rapid leukaemia cytoreduction of induction therapy may allow the immune system to exert sufficient control as to sustain remission, or to achieve tumour elimination. In this study, we aimed to characterise circulating T lymphocytes and those from the bone marrow (BM) tumour microenvironment, of paediatric B cell acute lymphoblastic leukaemia (ALL) patients at diagnosis and throughout remission induction chemotherapy.
17 paediatric patients diagnosed with precursor B cell ALL at Alder Hey Children's Hospital between August 2014 and February 2015 were enrolled in this study. Diagnosis, risk group assignment and treatment were performed according to the UKALL2011 multicentre trials. Philadelphia chromosome positive B-ALL or ALL in patients with Down syndrome were excluded. This study was approved by Norfolk Research Ethics Committee (REC 14/EE/0211) and informed consent was obtained from patients and/or their legal guardians in accordance with the Declaration of Helsinki. 87 paediatric BM and peripheral blood (PB) tissue samples were collected from enrolled patients at time points co-ordinating with treatment protocol (diagnosis, and induction days 8 and/or 15 and 29). CD4+ and CD8+ T cells, invariant natural killer T cells (iNKTs), cytokine induced killer T cells (CIKs) and γd T cells were stained with fluorochrome conjugated monoclonal antibody panels to allow subset analysis and activation state determination by six-colour flow cytometry.
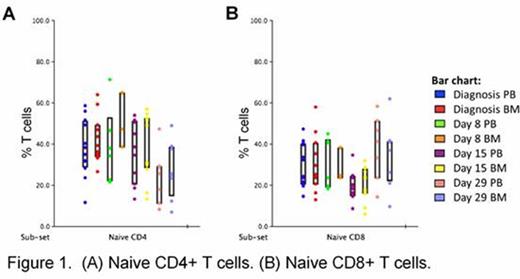
The relative frequencies of all major T cell populations; CD4+, CD8+, iNKTs, CIKs and γd T cells, were similar in PB and BM aspirate at diagnosis. CD4+ cells accounted for approximately 53% of circulating PB T cells and 47% of BM T cells. CD8+ cells accounted for approximately 38% of circulating PB T cells and 42% of BM T cells, the remaining 9% of circulating T cells and 11% of BM T cells were made up of combined iNKTs, CIKs and γd T cells. Naïve CD4+ and naïve CD8+ cells dominated both the circulating PB and BM T cell pools at diagnosis and throughout induction chemotherapy (Figure 1). Naïve CD4+ cells accounted for approximately 42% of circulating T cells and approximately 36% of BM T cells at diagnosis (Figure 1A). While naïve CD8+ cells accounted for approximately 21% of circulating T cells and 20% of BM T cells at diagnosis (Figure 1B). No significant changes in the relative frequencies of naïve CD4+ and CD8+ T cells were observed through induction days 8 and 15, although by induction day 29 the relative frequency of activated naïve CD4+ cells (HLA-DR+) was increased in the BM T cell pool compared to circulating naïve CD4+ cells in PB.
T cell reconstitution following induction chemotherapy may have important implications in childhood leukaemia, especially if recovering T cells are able to mediate anti-leukaemia effects. Rapid lymphocyte recovery has been associated with improved survival and is accounted for by increasing numbers of T lymphocytes following induction therapy in paediatric ALL. Whether anti-leukaemia T cell cytotoxicity develops following induction therapy remains to be elucidated although there is the potential for a cytotoxic response to develop in both CD4+ and CD8+ T cell subsets.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal