Abstract

Background: Tyrosine kinase inhibitors (TKIs) have dramatically improved outcomes in patients with Ph+ CML, in part by allowing achievement of sustained, low levels of BCR-ABL1 transcripts (quantified on the International Scale [IS]). TFR studies (eg, STIM, ENESTfreedom) evaluate whether some of these patients can stop TKI therapy and maintain a therapeutic molecular response off treatment. Here, we evaluated BCR-ABL1IS transcript levels of patients treated with frontline NIL 300 mg twice daily or IM 400 mg once daily for ≥ 1 year to predict time to TFR eligibility according to the ENESTfreedom trial criteria.
Methods: A statistical model was developed to predict the probability of future premature treatment discontinuation (due to adverse events, progression, or suboptimal response) and BCR-ABL1IS transcript levels at any time point after 1 year of treatment. The 5-year data from the ENESTnd clinical trial, in which BCR-ABL1IS transcript levels were assessed every 3 months, were the basis for this model. Probabilities of premature treatment discontinuation were modeled using parametric survival methods; early molecular response (EMR; BCR-ABL1IS ≤ 10% at 3 months) status was used as a predictor. For patients remaining on treatment, a second-order Markov chain model was used to predict probabilities of BCR-ABL1IS transcript levels being in each of 5 clinically relevant categories (≤ 0.0032% [MR4.5 ], > 0.0032% to ≤ 0.01% [MR4 ], > 0.01% to ≤ 0.1%, > 0.1% to ≤ 10%, and > 10%) at any time point after 1 year of therapy. Probabilities were a function of EMR status, the proportion of previous BCR-ABL1IS observations at or below MR4, and BCR-ABL1IS categories from the previous 2 assessments. A simulated cohort of 1000 patients was created to match the distribution of EMR status and BCR-ABL1IS categories in the first year of therapy in each of the trial populations (NIL or IM). Premature treatment discontinuation and BCR-ABL1IS categories were randomly drawn at each 3-month interval based on their corresponding predicted probabilities. Time to eligibility criteria for TFR was defined as: last BCR-ABL1IS assessment of MR4.5, none of the prior 3 assessments worse than MR4, and no more than 2 of the prior 3 assessments between MR4 and MR4.5.
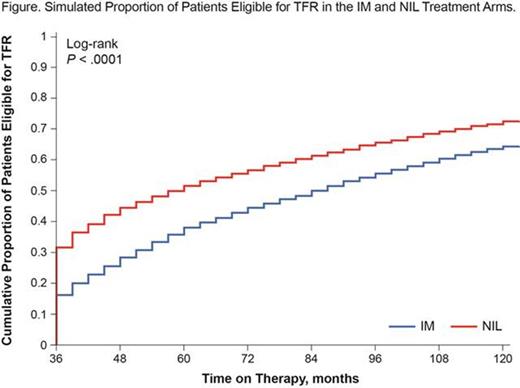
Results: For years 2 to 5 of the ENESTnd trial, the observed distribution of BCR-ABL1IS categories over time had reasonable agreement with the computer-simulated cohort. Simulation results (Figure) demonstrated that more patients on NIL than on IM were eligible for TFR by year 5 (52% vs 38%, respectively) and by year 10 (72% vs 64%, respectively; P < .0001 for both time points).
Conclusion: Patients in our simulated cohort received a minimum of 3 years of frontline treatment with NIL or IM prior to TFR eligibility evaluation, similar to the current consensus in clinical disease management. Treatment with NIL resulted in significantly more patients becoming eligible for TFR by all time points vs treatment with IM. These data suggest that TFR as a therapeutic goal may be more attainable with NIL than IM. Studies evaluating the duration of TFR are presently being conducted.
Mahon:Novartis: Consultancy, Honoraria; Bristol-Myers Squibb: Consultancy, Honoraria; ARIAD: Consultancy; Pfizer: Consultancy. Tran-Duy:Pharmerit International: Consultancy. Ray:Novartis Pharmaceutical Corporation/Rutgers University: Other: I am currently a fellow with Rutgers University, conducting my "field" experience at Novartis.. Mendelson:Novartis Pharmaceutical Corporation: Employment, Equity Ownership. Buchbinder:Novartis Pharmaceutical Corporation: Employment, Equity Ownership. Edrich:Novartis Pharma AG: Employment. Snedecor:Pharmerit International: Employment, Other: Institution received payment to conduct this study. Saglio:Pfizer: Consultancy, Honoraria; ARIAD: Consultancy, Honoraria; Bristol-Myers Squibb: Consultancy, Honoraria; Novartis Pharmaceutical Corporation: Consultancy, Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal