Abstract
The mechanisms leading to the establishment of the memory T-cell pool in humans remain to be fully elucidated. Murine models of memory T-cell generation propose an irreversible differentiation process from naïve to memory and effector cells. T-memory stem cells (TSCM) have been recently described in humans as constituting a key precursor subset of memory cells along this pathway.
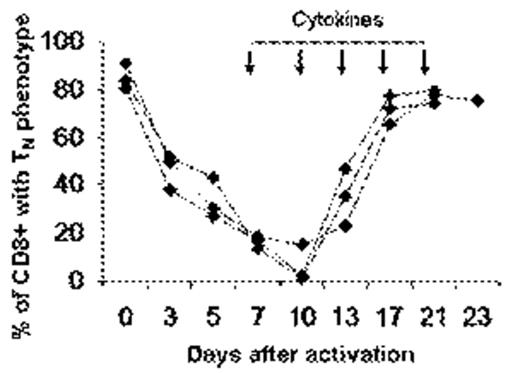
To study the generation of memory T-cells, primary naive T-cells (TN) derived from human cord blood (CB) were exposed to different activating stimuli, and then incubated with various cytokine combinations, and monitored throughout for phenotypic and functional changes. Between 3 to 9 days after antigen activation, the proportion of CD45RA+/CCR7+ CD8+ TN cells dropped from 86.64±5.84 (mean±1SD, n=50) to less than 20% (10.36±6.23). Specific cytokines mixtures were then added to the cultures and within 13 to 28 days after initial activation CD8+ cells re-expressed CD45RA, reverting back to a TN-like phenotype, characterized by the co-expression of CCR7, CD62L, CD27 and CD45RA, and loss of CD45RO. These constituted 71%±11.68 (1SD n=50) of the CD8+ T-cell population. The kinetics of 3 representative cultures are shown in Figure 1.
Recently differentiated central memory (TCM) and effector memory (TEM) CD8+ T-cells could be both induced to revert to a naïve-like phenotype (TNrev). Functional analysis showed that compared to TN, CD8+ TNrev displayed higher proliferative capacity upon antigenic stimulation, differentiated more quickly into progeny memory cells and acquired cytolytic effector function. CD8+ TNrev also had the ability to undergo several cycles of activation and differentiation whilst retaining the ability to revert back to TNrev.
CD8+ TNrev had an extended phenotype almost identical to TSCM, including the expression of CD95, and differed from TN in the expression of CXCR3 and Integrin b7. However, the expression of the latter markers and of CD95, became virtually undetectable following prolonged steady state culture and/or after modifying the cytokine milieu.
Here we describe for the first time the capacity of CD8+ TCM and TEM cells that have recently differentiated from primary TN to revert back to a naïve-like phenotype. CD8+ TNrev are functionally and phenotypically indistinguishable from TSCM, but with further changes to the cytokine milieu, they reacquire, with time, a phenotype resembling TN. TN Reversion may represent a way for the immune system to maintain a pool of antigen-experienced CD8+ T-cells with naïve characteristics, with implications for current models of memory T-cell generation. We demonstrate how understanding the T-cell differentiation process made possible the large scale in vitro generation of TSCM-like cells that can be used for adoptive immunotherapy strategies.
Kinetics of phenotype reversion following anti-CD3/IL2-activation of CB T-cells. When the percentage of TN cells dropped below 20%, cytokines were added and cells were monitored for phenotypic changes. Results of 3 representative experiments out of 50.
Kinetics of phenotype reversion following anti-CD3/IL2-activation of CB T-cells. When the percentage of TN cells dropped below 20%, cytokines were added and cells were monitored for phenotypic changes. Results of 3 representative experiments out of 50.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal