Abstract
Introduction
Numerous genetic abnormalities (including chromosomal translocations, deletions or amplifications, mutations) affect treatment outcomes in multiple myeloma (MM) and explain the heterogeneity in prognosis of MM patients with similar disease and host factors. Hyperdiploidy is associated with favorable outcomes, in contrast to del 17p, del 13, t(4;14), and gain of 1q2, which are associated with unfavorable outcomes. There is conflicting data about the role of overlap in cytogenetic and molecular abnormalities and their impact on prognosis. We sought from our study to determine the influence of overlapping genetic abnormalities in MM patients who received frontline autologous stem cell transplantation (ASCT) consolidation therapy.
Methods
Between January 2009 and January 2016, we included all consecutive newly diagnosed MM patients who underwent frontline ASCT at The University of Texas MD Anderson Cancer Center. All adult patients (≥18 years) who received high-dose melphalan conditioning regimen and had available conventional cytogenetics and interphase fluorescence in situ hybridization (FISH) studies at diagnosis were eligible. Patients with primary refractory and relapsed disease were excluded. Hyperdiploidy was defined as any extra copy of one or more of the odd chromosomes. High-risk genetic abnormalities are defined presence of del 17p, del 13, t(4;14), and/or 1q21 gain (detected by FISH and/or conventional cytogenetics). Primary and secondary endpoints were progression-free survival (PFS) and overall survival (OS), respectively. Survival estimates were calculated by Kaplan-Meir method, and Cox proportional hazards regression analysis was used to assess the predictors of PFS on univariate and multivariate analysis.
Results
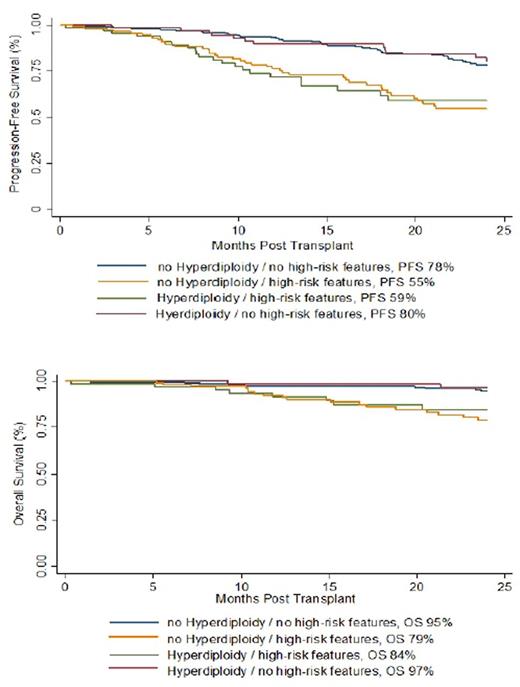
A total of 494 patients (57% males, 43% females) with a median age of 61 years (range, 33-80) were eligible and included in final analysis. 154 patients (31%) were identified to have any hyperdiploidy and 189 patients (38%) to have any high-risk abnormality. A total of 84 patients (17%) had hyperdiploidy without any high-risk feature and 121 patients (25%) had a high-risk cytogenetic abnormality without hyperdiploidy. With a median follow up of 27 months (range, 1-76) the 2-year PFS and OS of all study group were 71% and 90%, respectively. Among patients with any hyperdiploidy, the 2-year PFS and OS were 72% and 92%. In contrast, for patients with any high-risk genetic feature, the 2-year PFS and OS were 56%and 81%. Acquisition of any high-risk genetic abnormality, age >55 years, and international staging system (ISS) stage III were associated with worse PFS in univariate analysis. Further stratification of patients according to overlapping genetic abnormalities showed that the co-existence of hyperdiploidy with any high-risk genetic abnormalities was associated with significantly worse PFS compared to hyperdiploidy without co-exisiting genetic abnormalities (2-year PFS 59% vs 80%, HR 2.9, p=0.003) (Figure). PFS in patients with co-existing hyperdiploidy and high-risk genetic abnormalities was comparable to that in patients with high-risk genetic abnormalities without hyperdiploidy (59% vs. 55%, HR 0.9, 95%CI: 0.6-1.7; p=0.9) (Figure). The effect of high-risk abnormalities on PFS persisted on multivariate analysis, and was reflected on OS as well (Figure).
Conclusions
Our findings confirm that the co-existence of hyperdiploidy and high-risk genetic features does not abrogate the poor prognosis of MM patients with associated high-risk genetic abnormalities at diagnosis. Patients with hyperdiploidy and high-risk genetic abnormalities have similar outcomes to high-risk MM patients without hyperdiploidy, and should be considered as high-risk patients to guide future risk-adaptive treatment prospective clinical trials.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal