Abstract
Introduction: thrombotic thrombocytopenic purpura (TTP) is a rare, life-threatening thrombotic microangiopathy, with a reported incidence of 2-6 cases/million/year and a 10-20% mortality. TTP is associated with the severe deficiency of the von Willebrand factor cleaving protease, ADAMTS13, due to congenital defects in the ADAMTS13 gene (congenital TTP) or to the development of autoantibodies against ADAMTS13. The Milan TTP Registry is a digital database developed and curated at the Angelo Bianchi Bonomi Hemophilia and Thrombosis Center, Fondazione IRCCS Ca' Granda Ospedale Maggiore Policlinico (Milan, Italy), including demographic, clinical and laboratory data of patients with a confirmed diagnosis of congenital or acquired thrombotic thrombocytopenic purpura (TTP).
Aims: to report the natural history of patients affected with acquired TTP enrolled in the Milan TTP Registry, including demographic, clinical and laboratory data.
Methods: acquired TTP patients enrolled in the Milan TTP Registry for an acute episode of TTP occurred between January 2002 and November 2015, and followed until May 2016, were included in this study. Acquired TTP was defined as the occurrence of thrombocytopenia and microangiopathic hemolytic anemia, in the absence of alternative causes. Patients with acquired TTP secondary to cancer and bone-marrow transplant were excluded from this analysis. Demographic and disease-related information were collected by a standardized clinical questionnaire. ADAMTS13-related measurements were performed in a centralized laboratory in Milan. Remission of an acute TTP episode was defined as two days after normalization of the platelet count (≥150 x 109/l).
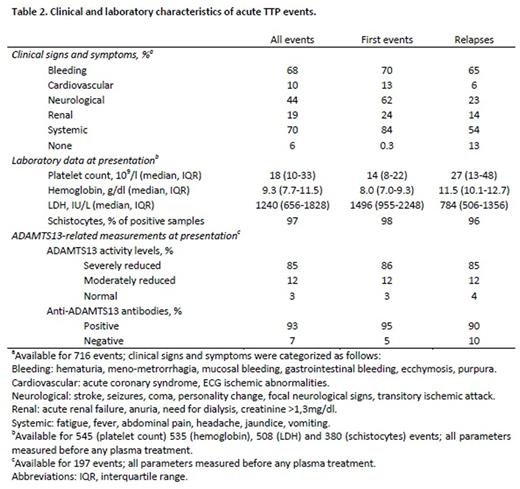
Results: 416 patients with a confirmed diagnosis of acquired TTP were included in the Milan TTP Registry, for a total of 837 acute events. Demographic, clinical and laboratory data are reported in Tables 1 and 2. The majority of patients were female (77%), of white ethnicity (98%) and Italian origin (82%). The median age at the first TTP episode was 40 years (interquartile range [IQR] 30-52). A history of other autoimmune diseases, cancer (non-active at the time of the TTP event), HIV infection and other diseases such as stroke, acute myocardial infarction and hypertension was reported in 34% of the patients. Among potential triggers of acute episodes (defined as occurring within three months before onset), infections were the most prevalent (22% of all patients), followed by the assumption of estroprogestinic drugs (5%). At TTP episode onset, systemic, bleeding and neurological signs and symptoms were present in 70%, 68% and 44% of acute events, respectively, whereas a lower prevalence of renal (19%) and cardiovascular (10%) signs and symptoms was observed. Clinical characteristics and hematologic laboratory parameters were less severe in relapsing episodes compared with first episodes (Table 2). Almost all acute events were treated by plasma exchange procedures and steroids. Rituximab therapy was administered in 12% of acute TTP episodes. In 197 acute TTP episodes with available ADAMTS13-related measurements at baseline, ADAMTS13 activity was severely reduced (<10%) in 85%, moderately reduced (10% to lower limit of normal range) in 12% and normal in 3% of events, respectively. Anti-ADAMTS13 antibodies were positive in 93% of acute TTP. The TTP-related mortality in our population of acquired TTP patients was 5%. In survivors, the median time to remission was 13 days (IQR 7-24, n=313), shorter for relapses than first events (median difference 8 days, 95% confidence interval 5-11). In 348 patients who survived the first TTP episode and had a follow-up period of at least 6 months, 178 (51%) experienced only one episode of acquired TTP, whereas 170 (49%) developed a recurrent form of the disease. This results might be overestimated due to the tertiary nature of our center.
Conclusions: acquired TTP is a rare, potentially fatal disease, which may require a long hospitalization and presents in a recurrent form in nearly half of the patients. Digital registries, such as the Milan TTP Registry, represent a powerful and necessary tool to systematically collect epidemiologic, clinical and laboratory data which may ultimately improve our understanding and management of acquired TTP. Our registry, in collaboration with those of other international centers, might answer the most relevant questions in this field.
Peyvandi:Novo Nordisk: Other: research funding paid to Luigi Villa Foundation, Research Funding, Speakers Bureau; CSL Behring: Speakers Bureau; SOBI: Speakers Bureau; Biotest: Other: research funding paid to Luigi Villa Foundation, Research Funding, Speakers Bureau; Ablynx: Membership on an entity's Board of Directors or advisory committees, Other: research funding paid to Luigi Villa Foundation, Research Funding; Octapharma: Consultancy; Alexion: Other: research funding paid to Luigi Villa Foundation, Research Funding; Bayer: Speakers Bureau; Grifols: Speakers Bureau; LFB: Consultancy; Kedrion Biopharma: Consultancy, Other: research funding paid to Luigi Villa Foundation, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal