Abstract

Introduction: As published by Naik both in single-institution and multi-institution data, adults with sickle cell disease have a baseline hypercoagulable state in addition to traditional risk factors. In pediatrics, venous thromboembolism (VTE) is often overlooked because of the low incidence in children. In addition, there is no unified consensus that supports the use of prophylaxis or screening of the pediatric population at risk. Although the incidence of VTE in the general pediatric population is low, there may be those children, including those with sickle cell disease (SCD), who would benefit from VTE prophylaxis.
Objectives: Estimate the contribution of sickle cell disease to rates of VTE in adolescents and young adults, compare rates of prophylaxis in the pediatric units versus the adult units, and determine risk factors associated with VTE occurrence in a hospital serving a large population with sickle cell disease.
Methods: This study was a retrospective chart review of patients aged 14-25 admitted to the Pediatric Intensive Care Unit (PICU), to the Pediatric Sickle Cell Service, and to the Medical Intensive Care Unit (MICU) between the dates of April 1, 2014 and April 30, 2015. Anyone older than age 18 admitted to the PICU or Pediatric Sickle Cell Service was considered part of pediatrics. Charts were searched manually for incidence of VTE during hospitalization, risk factors for developing VTE, history of prior VTE, use of therapeutic anticoagulation, and prophylaxis use and type. Data was analyzed using a multivariate regression analysis.
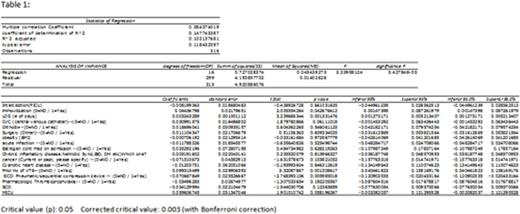
Results: In the 13-month period, 251 AYA admissions (108 to the PICU and 143 to the Sickle cell Unit) and 67 MICU admissions were reviewed. 66/67 (99%) MICU patients received either chemical or mechanical prophylaxis against VTE compared to 9/251 (3.5%) children. Event rates of VTE were 3/67 (4.5%) in MICU and 2/251 (0.8%) in the Pediatric units - 1 in the Sickle Cell unit and 1 in the PICU. In the pediatric units, none of the patients receiving prophylaxis had an acute VTE and both patients with VTE were not on prophylaxis. Both adolescents had central lines in place at the time of VTE occurrence. Of the MICU events, one patient had SCD and was taking systemic anticoagulation for previous VTE but had a central line in place. The other MICU patients both had infections, one with Crohns Disease and the other with a central line placed (table 2). The multivariate regression analysis demonstrated that prior history of a VTE and prolonged LOS were positively correlated with increased risk for VTE, both with a confidence interval of 95% and a corrected critical value of 0.003 using the Bonferroni correction (table 1).
Conclusion: Of the 5 VTE events in the 318 AYA hospitalizations reviewed, two (40%) were in patients with SCD (ages 17 and 24). As expected, overall VTE incidence in AYA is low in our institution, and appear to be associated with heightened inflammation and central line placement. VTE events in the pediatric unit were in patients not receiving any prophylaxis, which is not surprising when VTE prophylaxis orders were rare on the pediatric units (3.5%). In both our pediatric unit cases, prophylaxis may have prevented the VTE. A consensus protocol to identify high risk children is being implemented and refined with ongoing data collection. The data from our hospital AYA population enriched in SCD reinforces previous data from adult sickle cell populations that the magnitude of VTE risk attributable to SCD can be high. SCD probably should be among the screening criteria when deciding VTE prophylaxis in hospitalized adolescents.
Hsu:Purdue Pharma: Research Funding; Gerson Lehman Group: Consultancy; Hilton Publishing: Consultancy, Research Funding; Pfizer: Consultancy, Research Funding; Astra Zeneca: Consultancy, Research Funding; Eli Lilly: Research Funding; Sancilio: Research Funding; Centers for Medicare and Medicaid Innovation: Research Funding; EMMI Solutions: Consultancy; Mast Therapeutics: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal