Abstract
Introduction. Bone marrow (BM) aspiration is a routine test for hematologists, but it is a painful procedure for the patients. Since BM cells primarily stay in BM, not in the peripheral blood, BM analysis is an unavoidable procedure to diagnose hematological disorders. However, we previously reported that BM cells produce exosomes and such exosomes are released into the blood stream. Thus, by quantifying BM-cell specific mRNAs in plasma exosomes, we demonstrated that the condition of the BM could be assessed by using peripheral blood in patients with BM transplantation (Aoki et al. Clin Chem 60:675, 2014, ref. 1). Here we applied this analysis to much wider population of hematological disorders.
Materials and Methods. According to the Institutional Review Board (IRB)-approved procedure, a total 231 plasma samples from either peripheral blood or BM aspirate were collected. Two hundred mL of plasma were used to isolate exosomes followed by poly(A)+ mRNA isolation and cDNA synthesis as described previously (ref. 1). The resultant cDNA was used to amplify 10 different BM cell-specific mRNAs as well as 2 housekeeping genes, beta-actin (ACTB) and glyceraldehyde 3-phosphate dehydrogenase (GAPDH), by real time PCR (ref. 1). Using 10 fold dilution of 107 copies of ACTB and GAPDH DNA, cycle threshold was converted to copies/mL plasma.
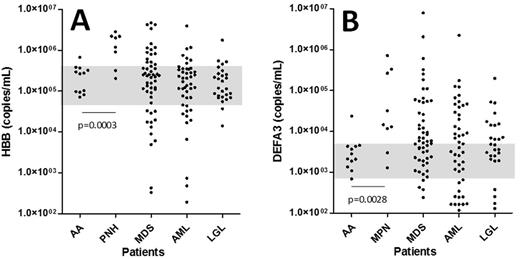
Results. We selected 12 aplastic anemia (AA) as a model of hypoplastic BM, 8 paroxysmal nocturnal hemoglobinuria (PNH) and 9 myeloproliferative neoplasms (MPN) as a model of hyperplastic BM for erythroid and myeloid cells, respectively. Then we analyzed various diseases including 53 myelodysplastic syndromes (MDS), 46 acute myeloid leukemia (AML), 27 large granular lymphocytic leukemia (LGL), etc., without subclassification of disease subtypes and staging. As shown in Fig. 1A, the levels of hemoglobin B (HBB) mRNA (copies/mL plasma) in PNH (each dot represents a single patient) were significantly higher than those of AA (p=0.0003) (AA area was blanketed), indicating erythroid hyperplasia during the hemolytic phase of the diseasese. Interestingly, MDS and AML demonstrated highly heterogeneous populations over PNH levels and much less than AA. The levels of myelocyte marker defensin A3 (DEFA3) mRNA in MPN were significantly higher than those of AA (p=0.0028) (Fig. 1B), indicating myeloid hyperplasia in MPN. Patients with MDS, AML, and LGL also showed highly heterogeneous populations, suggesting myeloid hypo- and hyperplastic BM dependent on the subtypes and stages of diseases.
Conclusion. This study indicates that plasma exosome mRNAs will become an interesting tool for hematologists to asses BM condition by use of a blood test. Although we only showed HBB and DEFA3 data in Fig. 1, this assay is a powerful platform for the biomarker discovery of various hematological disorders.
Quantification of erythroid (HBB) (A) and myeloid (DAFA3) (B) marker mRNAs in plasma exosomes. Each dot represents a single patient. Blanket is the area of AA.
Quantification of erythroid (HBB) (A) and myeloid (DAFA3) (B) marker mRNAs in plasma exosomes. Each dot represents a single patient. Blanket is the area of AA.
Mitsuhashi:NanoSomiX, Inc.: Employment; Hitachi Chemical America Co., Ltd.: Consultancy. Murakami:Hitachi Chemical America Co., Ltd.: Employment.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal