Abstract
Background:
One of the main challenges in the treatment of chronic myeloid leukemia (CML) is the selection of second line therapy. While Nilotinib (NILO) and dasatinib (DASA) are available for use as second line treatment for several years, few evidence-based data is available on how physicians make decisions on the use of one drug over another.
Aim:
We performed a pilot study to describe which factors physicians consider most relevant when deciding therapy with either NILO or DASA in patients who previously failed or were intolerant to Imatinib (IMA) therapy.
Patients and methods:
Analyses are based on a sample 67 CML patients, recruited as part of a larger international study, who switched from IMA therapy to either NILO (N=36; 53.7%) or DASA (N=31; 46.3%). Patients had to be in second line treatment for at least three months to be eligible for this analysis. Also, in all participating centers, NILO and DASA should have been equally available for use. There were 15 physicians involved in the management of these patients and they were asked to complete an ad-hoc questionnaire investigating reasons based on which they made the decision to either use one drug over another. All questions were phrased as follows: "To what extent have the following issues been determinant to make a decision on which agent (NILO or DASA) to use for this patient?" All answers were rated on a four point likert-scale (ie, not at all, a little, quite a bit and very much). Items investigated were: 1) accessibility of the drug in the hospital; 2) cost of drug; 3) patients' comorbidities; 4) patients' age; 5) patients' personality profile; 6) discussion with patients about Pro and Cons; 7) different treatment schedule of drugs; 8) type of mutation during IMA therapy. Physician characteristics were also collected and analyzed. Other treatment-related variables were investigated such as main reason for changing of TKI (IMA intolerance or resistance), high grade adverse events (AEs) experienced during IMA treatment, or previous duration of IMA therapy.
Results:
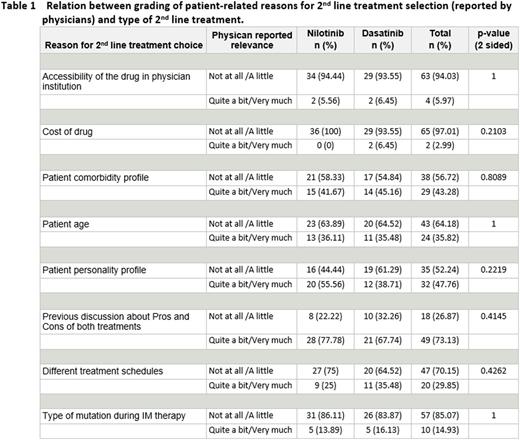
Physicians' experience in treating CML patients was on average 14 years (range 4-32). Patient median age at the time of treatment switch was 47 and 55 years in the NILO and DASA group, respectively. Median time of duration of IMA therapy, before receiving second line therapy, was 1.4 years and 3.3 years for those who switched respectively to NILO or DASA. No differences existed between groups with regard to reasons for switching from IMA therapy (intolerance or resistance) or AEs reported with previous IMA therapy. The top issue considered as most relevant when making the decision was previous discussion with patients on advantages and disadvantages of drugs, being reported as "quite a bit" and "very much" important in 73% of evaluations. Cost of the drug was not considered relevant at all, in the selection of which drug to use, in 97% of the 69 evaluations considered. Also, type of mutation during IMA therapy was considered as of negligible relevance for the decision, but it should not be overlooked that mutations were detected in few patients. Patient's comorbidity or personality profile was quoted as a 'quite a bit' or 'very much' relevant reason for the selection of 2nd line TKI, respectively in 43% or 48% of all questionnaires. Low relevance was assigned to patient age and different treatment schedule: 'not at all' or 'a little' relevance was reported respectively in 64% and 70% of all questionnaires. The analysis of physician-reported grading distribution according to the type of second line treatment did not show any significant difference (no reason lead to a preferential selection of one drug), see table 1.
Conclusions:
No differences were detectablein selected factors, driving the decision to switch either to NILO or DASA when physicians consider switching from first line IMA therapy. Also, type of TKI selection does not seems to be guided by only one factor. Further research is needed to elucidate on potential reasons underlying clinical decision making in 2nd TKI selection.
Efficace:Seattle Genetics: Consultancy; TEVA: Consultancy, Research Funding; Lundbeck: Research Funding; Bristol Myers Squibb: Consultancy. Rosti:Pfizer: Consultancy, Honoraria, Speakers Bureau; Novartis: Consultancy, Honoraria, Speakers Bureau; BMS: Consultancy, Honoraria, Speakers Bureau; Ariad: Consultancy, Honoraria, Speakers Bureau. Breccia:Celgene: Honoraria; Pfizer: Honoraria; Novartis: Consultancy, Honoraria; Bristol Myers Squibb: Honoraria; Ariad: Honoraria. Baccarani:Novartis: Consultancy, Honoraria, Speakers Bureau; Pfizer: Consultancy, Honoraria, Speakers Bureau; BMS: Consultancy, Honoraria, Speakers Bureau; Ariad: Consultancy, Honoraria, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal