Abstract
The major complication of severe aplastic anemia is clonal evolution, defined as any new cytogenetic abnormality or progression to MDS/AML, which occurs in about 15% of SAA patients, usually many months to years after the diagnosis. Eltrombopag, a thrombopoietin receptor agonist, appears capable of stimulating hematopoietic stem cell proliferation in patients with bone marrow failure. Addition of eltrombopag to standard immunosuppressive treatment (IST) with horse antithymocyte globulin (hATG) and cyclosporine (CsA) markedly increases hematologic response rates in treatment-naive SAA, with overall response rates up to 90% and complete response rates approaching 60% (Townsley DM et al, ASH 2015, clinicaltrials.gov NCT01623167). In comparison, IST alone achieves 60% overall response rates, of which 10% are complete (Scheinberg Blood 2012). Somatic mutations in myeloid cancer candidate genes are present in one-third of patients after IST alone (Yoshizato T et al, NEJM 2015). Specific subsets of mutations were associated with clinical outcomes: a group including ASXL1 and DNMT3A with a poor response to IST, inferior survival, and clonal evolution, while BCOR and PIGA were associated with good response and favorable outcomes. Monosomy 7, the most prevalent cytogenetic abnormality defining clonal evolution, can develop in the absence of gene mutations, underscoring the non-determinative and complex role mutations play in clonal evolution (Dumitriu B et al, Blood 2015). Of patients with disease refractory to IST who were subsequently treated with single-agent eltrombopag, 19% (8/43) developed cytogenetic abnormalities, usually within the first year of treatment, but only rarely with morphologic changes consistent with MDS/AML (Desmond R et al, Blood 2014). The frequency of somatic mutations following treatment with eltrombopag added to IST in treatment-naïve SAA patients is unknown.
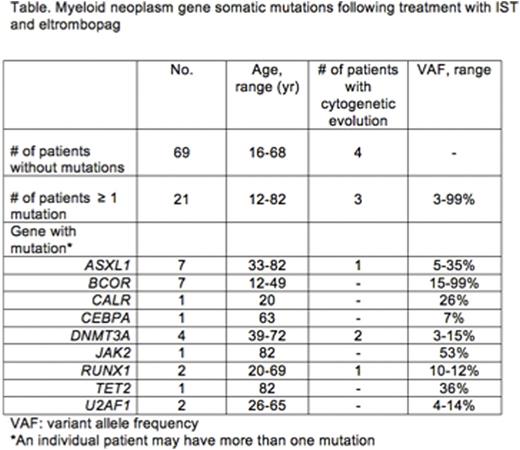
We used amplicon-based next-generation sequencing to assess mutations in 54 candidate genes recurrently mutated in myeloid neoplasms. Bone marrow cells of 90 subjects who had been treated with IST/eltrombopag were obtained at 6 months following treatment initiation, or at the time of clonal evolution. At least one detectable mutation was identified in 21 (23%) subjects. All 21 patients had exhibited a hematologic response to treatment by 6 months; of those patients with somatic mutations, 14 of 19 (74%) had a complete hematologic response. In comparison, of the 69 patients who lacked mutations, 20 (29%) had a complete response. Nine different genes were mutated in total, with the most frequent genes being ASXL1 and BCOR. BCOR was associated with more robust responses (6 of 7 had a complete response) and younger age (range 12 - 49 years; Table). One subject with a longstanding history of JAK2-positive essential thrombocytosis and myelofibrosis at baseline had two additional mutations detectable following treatment, TET2 and ASXL1.
With a median follow up of 21 months, clonal cytogenetic evolution occurred in 7/90 (8%) subjects. Three of seven patients also had a mutation in a myeloid cancer gene (two with DNMT3A and one with ASXL1/RUNX1); in the other four, no somatic mutation was detected, either at the 6-month time point or at time of cytogenetic evolution. Four patients had monosomy or partial deletion of chromosome 7: one patient had complex (t(3;3)(q21;q26), -7), one patient had deletion 13q that later disappeared, and one patient had trisomy 6 and trisomy 15 in 2 metaphases. The patient with complex cytogenetics did not have somatic mutations detected at evolution, and she later died due to relapsed AML following transplant.
The rates of somatic mutations in myeloid cancer genes and of cytogenetic evolution in patients treated with IST/eltrombopag do not appear to be higher than we and others have reported in aplastic anemia treated with standard IST, without eltrombopag (Kulasekararaj AG et al, Blood 2014). The distribution of genes mutated and the allelic frequency of these mutations also were similar to patients treated with standard IST. These results suggest that the benefits of a higher response rate and quality of response associated with the addition of eltrombopag to IST for the initial treatment of SAA are not associated with a higher risk of clonal progression.
Townsley:GSK/Novartis: Research Funding. Cooper:GSK/Novartis: Research Funding. Winkler:GSK/Novartis: Research Funding. Scheinberg:Novartis: Consultancy, Speakers Bureau. Rios:GSK/Novartis: Research Funding. Weinstein:GSK/Novartis: Research Funding. Desierto:GSK/Novartis: Research Funding. Fernandez Ibanez:GSK/Novartis: Research Funding. Dunbar:GSK/Novartis: Research Funding. Ma:Neogenomics Laboratories: Employment. Albitar:Neogenomics Laboratories: Employment, Equity Ownership. Young:GSK/Novartis: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal