In this issue of Blood, Banerjee et al show that circulating platelets engulf thrombotic proteins and then recycle them during platelet activation and thrombosis.1
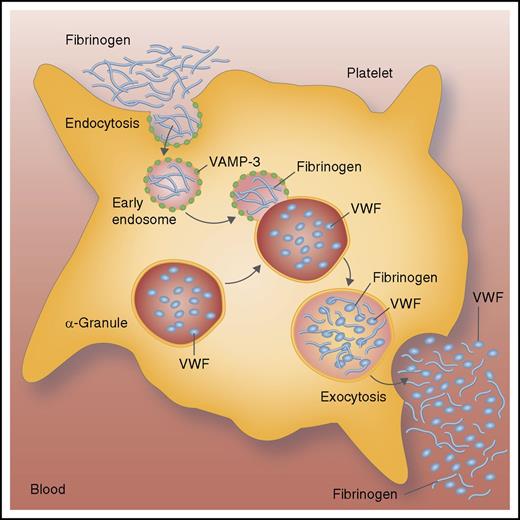
Platelets take up and release fibrinogen. Platelets engulf fibrinogen from the blood and load it into endosomes through the process of endocytosis. Endosomes containing fibrinogen fuse with granules containing VWF. The protein VAMP-3 facilitates this membrane fusion. During platelet activation, platelets release fibrinogen and VWF into the blood to promote platelet aggregation and thrombosis. Professional illustration by Somersault18:24.
Platelets take up and release fibrinogen. Platelets engulf fibrinogen from the blood and load it into endosomes through the process of endocytosis. Endosomes containing fibrinogen fuse with granules containing VWF. The protein VAMP-3 facilitates this membrane fusion. During platelet activation, platelets release fibrinogen and VWF into the blood to promote platelet aggregation and thrombosis. Professional illustration by Somersault18:24.
Platelet activation depends upon exocytosis, a process that involves platelet granules releasing their prothrombotic contents into the blood. Banerjee and colleagues now show that platelets rely on endocytosis as well. Endocytosis is the process of selectively packaging surface proteins into vesicles called endosomes.2 These early endosomes then transport their cargo to distinct locations inside the cell, such as late endosomes for degradation, or back to the plasma membrane, where they are recycled. (Endocytosis usually involves clathrin-coated vesicles, although surface cargo can also be engulfed by other types of vesicles without clathrin coats, such as caveolae or macropinosomes.)
Platelet endocytosis can store ligands and limit receptor signaling. Prior studies showed that platelets take up plasma proteins such as von Willebrand factor (VWF), fibrinogen, and albumin.3 These proteins are then delivered to α-granules, where they are stored and then subsequently released into the blood. Platelets also internalize platelet surface receptors such as the integrins GPIIbIIIa and GPIb and the purinergic receptor P2Y12.4,5 Presumably, engulfing integrins would limit platelet attachment to the vessel wall, and decreasing receptors such as P2Y12 would dampen platelet activation. However, the mechanisms of platelet endocytosis were previously unknown.
How do platelets ingest ligands like fibrinogen? In the current study, Banerjee and colleagues explored the pathway through which fibrinogen binds to GPIIbIIIa and then enters the platelet. They discovered, based on several key observations, that vesicle-associated membrane protein-3 (VAMP-3) plays a key role in platelet endocytosis (see figure). Platelets lacking VAMP-3 contain less fibrinogen than normal platelets. Without VAMP-3, platelets cannot take up and store fibrinogen within α-granules. Next, the scientists examined the functional effect of VAMP-3 upon receptor signaling and showed that VAMP-3 controls signaling of the thrombopoietin receptor, probably by regulating internalization and intracellular signaling. Finally, the research team showed that VAMP-3 also limits platelet spreading and clot retraction. Thus, VAMP-3 mediates platelet endocytosis.
How does endocytosis regulate platelet spreading? Others have shown that VAMP-3 endosomes regulate cell adhesion in other cells, such as monocytes.6 One possibility is that endocytosis removes surface receptors that activate the spreading process; another option is that endocytosis decreases the surface expression of integrins that themselves mediate spreading.7
These studies suggest that endocytosis plays several critical roles inside platelets:
First, endocytosis might permit platelets to take up prothrombotic and proinflammatory mediators that are not normally synthesized inside megakaryocytes; these messengers can then be stored and later released during activation. This raises interesting questions: What other plasma proteins are taken up into platelets through endocytosis? Are these proteins stored for later release, or are they degraded?
Second, endocytosis might regulate platelet signaling by delivering receptor–ligand pairs to the cell interior, much like Toll-like receptors and their ligands signal inside cells from vesicular compartments.
Third, endocytosis could modulate platelet interactions with other cells, such as platelets or neutrophils, by controlling the density of surface receptors.
What is the physiological relevance of endocytosis in platelets? The phenotype of VAMP-3 null mice has not been fully explored. Banerjee’s team show that VAMP-3 does not affect hemostasis and thrombosis. Does VAMP-3 affect innate immunity? Platelets release a variety of prothrombotic compounds, such as platelet factor 4 (CXCL4), and it is possible that platelets could take up and store chemokines through receptor-mediated endocytosis, subsequently releasing them to activate neutrophils and monocytes.8 Can VAMP-3 modulate adaptive immunity? Dendritic cells take up antigens into endosomes, where the antigens are processed, loaded onto major histocompatibility complex (MHC) class I, and recycled to the surface for antigen presentation. Perhaps platelets take up foreign antigens in a VAMP-3–dependent process or deliver vesicles with MHC class I to vesicles with antigens through VAMP-3.9 Finally, platelets undergo autophagy during starvation, consuming granule contents to generate energy, and VAMP-3 regulates membrane fusion in autophagy, so perhaps VAMP-3 regulates platelet autophagy.10 Further exciting studies will no doubt reveal the physiological significance of platelet-based endocytosis.
Conflict-of-interest disclosure: The author declares no competing financial interests.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal