Abstract
Background: Entospletinib (ENTO) is a selective spleen tyrosine kinase (SYK) inhibitor that has demonstrated evidence of efficacy and tolerability in chronic lymphocytic leukemia (CLL). The relationship between ENTO plasma exposures and clinical efficacy and safety were evaluated in relapsed/refractory (R/R) CLL subjects, following administration of 100, 200, and 400 mg twice daily (BID) in a Phase 2 Study (NCT01799889; study -0102).
Methods: ENTO pharmacokinetics (PK) and plasma exposures (Cmax, AUCtau, and Ctau) were characterized in 60 R/R CLL subjects using a population PK model developed from data collected in phase 1/2 clinical studies. ENTO plasma samples were collected on day 1, and day 8 (pre-dose,1.5 hours, and 4 hours post-dose), day 15, and day 22 (pre- and 1.5 hours post-dose) of cycle 1 and on day 1 (pre- and 1.5 hours post-dose) of subsequent 28 day cycles. Data for efficacy and safety endpoints were collected every 8 weeks (for cycles 1-6) followed by every 12 weeks (for cycles 7-18). Efficacy endpoints, including best overall response rate (BOR), progression-free survival (PFS), sum of products of the greatest perpendicular diameters (SPD) of index lesions, maximum change from baseline in lymphocytes, platelets, and hemoglobin were evaluated against ENTO plasma exposure (AUCtau and Ctau). Safety endpoints, including adverse events (all grades) of skin rash, infection, diarrhea, aspartate aminotransferase (AST), alanine aminotransferase (ALT), bilirubin (total), and neutrophils (maximum change from baseline) versus ENTO plasma exposure (AUCtau and Cmax) were evaluated.
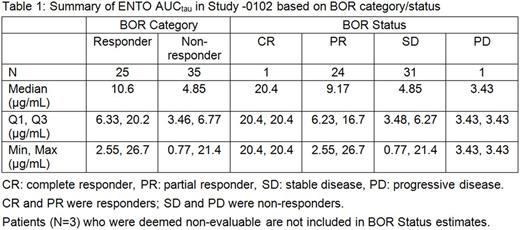
Results: A dose proportional increase in ENTO exposures was observed across 100, 200, and 400 mg BID regimen. A greater reduction in SPD and lymphocytes and an increase in platelets were associated with higher ENTO exposures. No exposure-response relationship was observed for PFS at Week 24 (i.e., there was an overlap in survival curves across quartiles of ENTO exposures) or hemoglobin. No association between ENTO exposures vs incidence of skin rash, infection, diarrhea, AST, ALT and neutrophils was observed. There was a slight trend toward elevated total bilirubin with higher ENTO exposures.
Conclusions:In R/R CLL subjects, following ENTO 100, 200, and 400 mg BID regimen, higher exposures corresponding to 400 mg BID are associated with greater efficacy (reduction in SPD lymphocytes and an increase in platelets). No clinically relevant relationship between ENTO exposures and safety, except total bilirubin, was observed. Overall, the analyses support ENTO 400 mg BID regimen as a starting dose for future phase 2 trials in CLL patients.
Gupta: Gilead Sciences, Inc.: Employment, Equity Ownership. Meng: Gilead Sciences, Inc.: Employment, Equity Ownership. Sharma: Gilead Sciences, Inc.: Employment, Equity Ownership. Ye: Gilead Sciences, Inc: Employment, Equity Ownership. Silverman: Gilead Sciences, Inc.: Employment, Equity Ownership. Abella: Gilead Sciences, Inc.: Employment, Equity Ownership. Mathias: Gilead Sciences, Inc.: Employment, Equity Ownership.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal