Abstract
Introduction:
The risk of thrombosis is markedly increased in patients with cancer and associated with increased morbidity and mortality. Clinical scoring systems are helpful to stratify cancer patients at a highest risk for thrombotic events. However, more precise risk prediction is needed, particularly for patients with cancers associated with intermediate thrombotic risk. In some, but not all studies, evidence of activation of coagulation proteases as measured using assays such as thrombin generation times, D-dimer levels, etc., has correlated with thrombotic risk, though these correlations are generally not robust. Recent studies have demonstrated that the contact activation pathway plays an important role in the development of pathologic thrombosis, though its role in cancer-associated thrombosis has not been clearly defined. However, unlike assays that measure prothrombin activation, limited options for assessing contact activation are available. To examine this question, we have developed a quantitative immunoassay for contact activation that assesses cleavage of high molecular weight kininogen (HK), a marker of contact system activation, in patient plasma. Circulating cleaved HK (HKa) has been found in plasma in disorders such as hereditary angioedema, and our studies in mice demonstrate that HK is quantitatively cleaved within murine tumors.
Methods:
To detect circulating HKa, one microliter of plasma was analyzed using SDS-PAGE on a 4-12% gel under reduced conditions. Following transfer to low-fluorescence PVDF membranes, the sample was immunoblotted using an affinity-purified rabbit polyclonal antibody raised to a peptide encompassed by the C-terminal region of the HK heavy chain (CQPLGMISLMK). Bound antibody was detected using an IRDye® 800CW-labeled goat anti-Rabbit secondary antibody followed by band quantification using a LI-COR Odyssey imaging system and Image StudioTM Lite Software. This assay detects both intact kininogen (MW ~ 120 kD) and the cleaved kininogen heavy chain (MW ~ 50 kD). To control for different levels of kininogen in individual patients, cleaved kininogen was quantified by estimating the relative amount of kininogen heavy chain as a percentage of uncleaved kininogen using LI-COR technology.
Results:
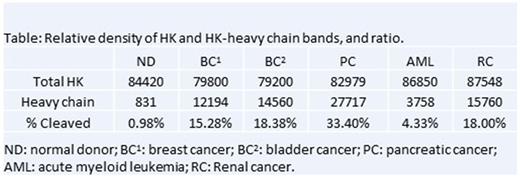
To date, we have used this assay to assess kininogen cleavage in 11 patients with several different cancers including breast cancer (N = 3), bladder cancer (N = 1), pancreatic cancer (N = 5), acute myeloid leukemia (N = 1), and renal cancer (N = 1). Patients with cancer demonstrated a greater percentage of cleaved HK in plasma than normal donor controls, in whom less than 1% of kininogen was present in the cleaved form. The greatest amount of cleavage was observed in pancreatic cancer, and was estimated at 33.4% (see Table).
Conclusion:
These studies provide evidence that the contact activation system is activated in many patients with cancer. Current effort is focused on identifying causes of variation in contact activation between cancers, and determining whether the extent of contact activation as determined by this quantitative assay correlates with or predicts the development of thrombosis in cancer patients.
Khorana: Halozyme: Consultancy, Honoraria; Sanofi: Consultancy, Honoraria; Pfizer: Consultancy, Honoraria; Bayer: Consultancy, Honoraria; Amgen: Consultancy, Honoraria, Research Funding; Leo: Consultancy, Honoraria, Research Funding; Janssen Scientific Affairs, LLC: Consultancy, Honoraria, Research Funding; Roche: Consultancy, Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal