Abstract
Background
CD47 is an immune checkpoint that binds signal regulatory protein alpha (SIRPα) and delivers a "do not eat" signal to suppress macrophage phagocytosis. Tumor cells, including T-cell lymphomas, frequently overexpress CD47 to escape immune surveillance. TTI-621 (SIRPαFc) is a fusion protein consisting of the CD47 binding domain of human SIRPα linked to the Fc region of human IgG1, designed to enhance phagocytosis and antitumor activity by blocking the CD47-SIRPα interaction between malignant cells and macrophages, and engaging activating Fcγ receptors. It is hypothesized that direct intralesional (IL) administration of TTI-621 may enhance both local and systemic antitumor activity.
Methods
A multicenter, open-label Phase 1 study is ongoing to characterize the safety, tolerability, pharmacokinetics, pharmacodynamics and antitumor activity of IL injections of TTI-621 (NCT02890368) in adult patients with relapsed/refractory (R/R) percutaneously accessible solid tumors and mycosis fungoides (MF) who have progressed on standard anticancer therapy or for whom no other approved therapy exists. The current report analyzed a cohort of R/R MF/Sézary syndrome (SS) patients enrolled by the data cut-off date. Patients received induction therapy consisting of a single IL injection (1, 3 or 10 mg), three 10 mg injections over one week, or six 10 mg injections over two weeks distributed across one to three lesions. The protocol was recently amended to enable weekly post-induction IL maintenance dosing and to explore IL TTI-621 administration in combination with subcutaneous pegylated interferon-α2a. Responses were evaluated using the Composite Assessment of Index Lesion Severity (CAILS) score a week after induction therapy and at later time points in some patients. Serial biopsies were collected to assess the impact of TTI-621 on the tumor microenvironment.
Results
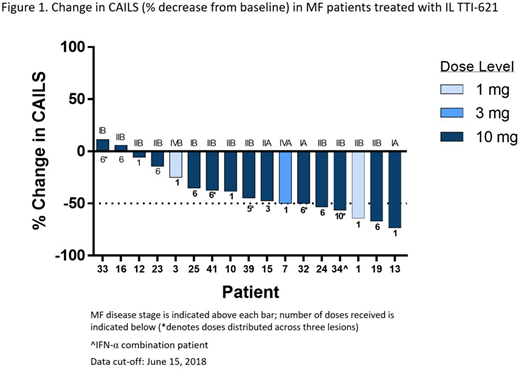
Twenty-two patients (16 M/6F, median age 65.5 years, range 32-85) were enrolled as of June 15, 2018. Primary diagnosis included MF (n=18), MF with transformation (n=3) and SS (n=1). Clinical stages included stage IA (n=2), IB (n=3), IIA (n=1), IIB (n=13), IVA (n=2) and IVB (n=1). Patients received a median of 3 prior systemic therapy regimens. Twenty-one patients were treated with TTI-621 monotherapy and one patient with TTI-621 in combination with pegylated interferon-α2a. Single and multiple IL injections of up to 10 mg TTI-621 have been well tolerated. The most frequently reported treatment-related adverse events (AEs) were chills (n=8), injection site pain (n=7) and fatigue (n=6). All treatment-related AEs were Grade 1 or 2 in severity. No treatment-related serious AEs or dose-limiting toxicity have been observed. CAILS scores for injected lesions obtained after the last injection were available for 17 patients: 15 (88%) had measurable improvement; 7 (41%) exhibited ≥50% decrease from baseline (Figure 1). Responses were rapid and in some patients occurred after a single injection of varying doses of TTI-621. CAILS scores continued to decrease in 5/5 (100%) monotherapy patients for which post-induction therapy assessments were conducted. Seven patients with reduced CAILS of varying degree (-14% to -67%) in the injected lesions had paired assessments available in adjacent, non-injected lesions. In 6/7 patients, a reduction in CAILS was observed in the non-injected lesions (-12% to -67%), indicating that TTI-621 could induce local-regional responses that were not confined to the site of injection. Additionally, one patient with transformed MF exhibited clear evidence of abscopal effects with rapid resolution of lesions on the abdomen, left flank/back and arms less than two weeks after receiving IL TTI-621 injections in lesions on the foot and leg. The only SS patient on study achieved a reduction in circulating Sézary cells after a single 3 mg local injection of TTI-621.
Conclusions
Preliminary data from this ongoing study indicate that IL TTI-621 administration is well-tolerated and has single agent activity in heavily pre-treated MF/SS patients across various disease stages. The rapid responses observed occurred in both injected and non-injected lesions indicating a local-regional effect with initial evidence of distant abscopal or systemic effects. Enrollment in this study is continuing to evaluate the impact of weekly maintenance dosing and further characterize the systemic effect and durability of responses.
Querfeld:Kyowa: Membership on an entity's Board of Directors or advisory committees; Acelion: Membership on an entity's Board of Directors or advisory committees; Bioniz: Membership on an entity's Board of Directors or advisory committees; Medivir: Membership on an entity's Board of Directors or advisory committees; Trillium Therapeutics: Membership on an entity's Board of Directors or advisory committees. Thompson:Trillium Therapeutics: Research Funding. Taylor:Trillium Therapeutics: Research Funding. Johnson:Trillium Therapeutics: Employment. Catalano:Trillium Therapeutics: Employment. Petrova:Trillium Therapeutics: Employment. Thompson:Trillium Therapeutics: Employment. Uger:Trillium Therapeutics: Employment. Shou:Trillium Therapeutics: Employment. Akilov:Kyowa Kirin: Consultancy; Pfizer: Research Funding; Seattle Genetics: Consultancy; Trillium Therapeutics: Research Funding; Actelion Pharmaceuticals: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal