Introduction: Andexanet Alpha (Coagulation factor Xa recombinant, inactivated Zh-zo; AA, Portola Pharmaceuticals) is a recombinant factor Xa decoy protein which is designed to reverse the effects of apixaban and rivaroxaban and is approved for the control of bleeding complications associated with their use. The molecular modification in this recombinant protein involves the substitution of serine active site by alanine and the removal of the gamma-carboxyglutamic acid (GLA) domain to restrict its assemblage into prothrombinase complex. Beside the reversal of the effects of anti-Xa agents AA is also reported to neutralize the biologic effects of heparin and related drugs. Assay dependent variations in the neutralization profile of various factor Xa inhibitors by andexanet has been recently reported https://doi.org/10.1177/1076029619847524. Since heparin and related drugs also mediate their biologic actions by inhibiting factor Xa via AT complexation, it is hypothesized that AA may also inhibit their biologic effects as measured in various laboratory assays. It is the purpose of this study is to compare the relative neutralization profile of heparin (UFH), a low molecular weight heparin, enoxaparin (E) and a chemically synthetic pentasaccharide, Fondaparinux (F) by AA.
Materials and Methods: API versions of UFH, E and F were commercially obtained in powdered forms and dissolved in saline at a working dilution of 1mg/ml. AA was dissolved in saline to obtain a 10mg/ml working solution. The anticoagulant profile of UFH, E and F was studied using the activated partial thromboplastin time (APTT) and thrombin time (TT) in a concentration range of 0 - 10 ug/ml in pooled human plasma. The anti-Xa and anti-IIa studies were carried out in amidolytic assays in the same concentration range. The thrombin generation inhibition was studied using calibrated automated thrombin generation systems (CAT, Diagnostica Stago). The effect of AA on the reversal of the anticoagulant and anti-protease and thrombin generation effects of each of these agents were studied by supplementing this agent at 100 ug/ml. The results are compared to determine the difference between pre and post AA neutralization settings.
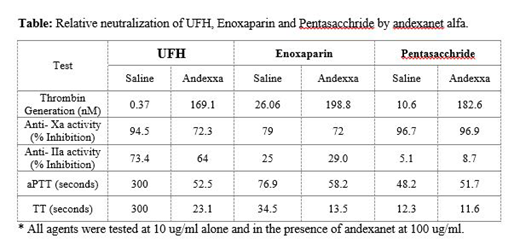
Results: All agents produce a concentration dependent effect in the anticoagulant and anti-protease assays with the exception of F which showed mild anticoagulant effects, and very weak anti-IIa actions and strong anti-Xa activity. In the anti-Xa assay the IC-50 for UFH was 2.1ug/ml (0.13 um), E 4.3 ug/ml (0.95 um) and F 0.7 ug/ml (0.41 um) upon supplementation of AA the IC50s for UFH was increased to 5 ug/ml (0.31 um) and for E 5 ug/ml (1.11 um). However, there was no neutralization of the anti-Xa effects of the F by AA and the IC50 remained the same for both pre and post andexxa studies. The anticoagulant effects of UFH as measured by aPTT and TT was strongly neutralized whereas E was only partially neutralized in the aPTT assay and almost completely neutralized in the thrombin time assay. At concentrations of up to 10 ug/ml F did not produced any significant anticoagulant effects, both in the presence and absence of AA. In the thrombin generation inhibition assays, UFH produced a complete inhibition of thrombin generation which was completely reversed by AA. Although both E and F produced strong inhibition of thrombin generation, AA did not completely neutralize these effects. The results are tabulated on table 1 for the studies carried out at 10 ug/ml of UFH, E and F.
Conclusion: These results indicate that AA is capable of differentially neutralizing anticoagulant and anti-protease effects of UFH in an assay dependent manner. However, AA is incapable of neutralizing the anti-Xa effects of E and F. This may be due to the relatively differential affinities of enoxaparin and fondaparinux AT complex to factor Xa rendering it inhibited in the presence of AA. These studies also demonstrate that the primary surrogate marker anti-Xa activity for measuring the activities of anti-Xa agents is not proportional to the anticoagulant and thrombin generation inhibitory effects of these agents. A global clotting assay may be a better indication of the biologic effects of these agents and their reversal by AA.
Tafur:Recovery Force: Consultancy; Janssen: Other: Educational Grants, Research Funding; BMS: Research Funding; Idorsia: Research Funding; Daichi Sanyo: Research Funding; Stago: Research Funding; Doasense: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal