Introduction: Since the publication of the International Guidelines (Borowitz, 2010; Illingworth, 2018), no study has assessed the long-term evolution of paroxysmal nocturnal hemoglobinuria (PNH) clones using high-resolution flow cytometry. The sole evaluation, performed by Sugimori et al, using a 2-color flow cytometry test, showed the disappearance of PNH clones in 24% of patients with bone marrow (BM) failure over 5 years (Sugimori, 2009). A diagnostic practice harmonization using high-resolution flow cytometry has spread in France since 2013 through an on-going inter-laboratory comparison program (Debliquis, 2015). Thus, our HPNAFC group has been able to initiate a French nation-wide multicenter prospective observational study.
Objective: We aimed to assess the evolution of PNH clones over a long term period using mostly high sensitivity test, which is required for minor clone assessment, with validated flow cytometry data.
Methods: All patients of any age with a PNH clone or GPI-deficient cells ≥0.01%, newly- or previously-diagnosed, detected in France from February 29th 2016, could be included in this Observatory, provided that the center had validated a PNH flow cytometry quality control. For each patient, the baseline assessment was always considered as the initial PNH clone detection, even if it occurred before the initiation of the Observatory. Thus, this strategy allowed the collection of cases with long-term follow-up. Referent cytometrist of each center included patients in the e-CRF available on the HPNAFC website providing clinical and biological information as well as flow cytometry raw data files. This study was approved by the national research ethics board.
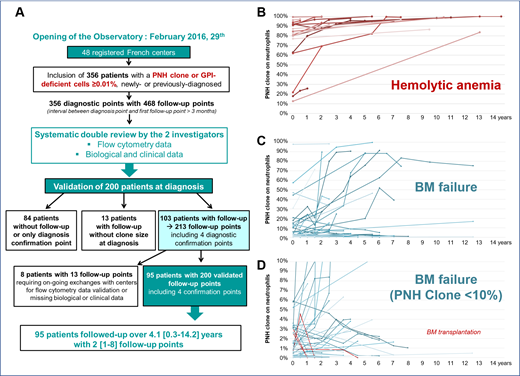
Results: As of July 15th 2019, 48 participating flow cytometry laboratories across France have enrolled 356 patients with a PNH clone or GPI-deficient cells ≥ 0.01%. All cases have been carefully reviewed by the 2 principal investigators, who both thoroughly re-examined flow cytometry data and the e-CRF filling that led to the update of roughly one third of the submitted files. This enabled the validation of 200 patients at diagnosis, the remaining 156 being ongoing. One hundred and three of the 200 validated patients displayed at least one follow-up point (more than 3 months apart from the diagnosis) with a clone size determined at diagnosis (see flow chart figure 1A). For 8/103 patients, exchanges with centers are still ongoing. Thus, we were able to assess the evolution of PNH clones of 95 patients with 2 [range: 1-8] follow-up points over a period of 4.1 [0.3-14.2] years, corresponding to 200 validated follow-up points. The patient median age at diagnosis was 40 years old [10-85] with 3 pediatric cases (<18y) and a M/F sex ratio of 0.86. Diagnoses were made between 2003 and 2018 with clinical information available in 97% of cases: 19 patients (20%) had hemolytic anemia and most patients (n=73, 77%) displayed BM failure including aplastic anemia (n=62), myelodysplastic syndrome (n=7) and unexplained cytopenia(s) (n=4). No case of thrombosis was included. All patients with hemolytic anemia showed an increasing clone size over time including the two who were not treated with eculizumab (Figure 1B; median size at diagnosis on neutrophils: 81.3% vs median size over 5 years: 96.3%). The median clone size at diagnosis for patients with BM failure was 1.5% on neutrophils with a very wide range [0.01-97.87], almost half of them being less than 1%. When comparing the diagnosis point with the latest follow-up point, PNH clone size increased in 37 patients, decreased in 16 of them and remained stable in 20 cases (Figure 1C, D). Nine of the 37 patients reached a PNH clone size above 50% and 4 of them received a treatment by eculizumab in a median delay of 5 years [1.5-6.0]. Interestingly, no patient showed spontaneous disappearance of PNH clones, pending the use of a high-resolution flow cytometry test. The only five patients with undetectable PNH clones (Figure 1D, red lines) were those who underwent BM transplantation.
Conclusion: This multicenter study based on robust flow cytometry analysis showed no disappearance of PNH clones, including minor ones, over a long period of time, regardless of the clinical manifestations, except for patients who underwent BM transplantation. Moreover, PNH clone size increased in half of patients with BM failure, justifying a long term PNH clone size monitoring, even in these patients.
Le Garff-Tavernier:Alexion: Consultancy, Honoraria. Pruvot Debliquis:Alexion: Honoraria; Takeda: Honoraria; Pfizer: Honoraria; Gilead: Honoraria; Genzyme: Honoraria. Socie:Alexion: Consultancy. Peffault de Latour:Alexion: Consultancy, Honoraria, Research Funding; Pfizer: Consultancy, Honoraria, Research Funding; Novartis: Consultancy, Honoraria, Research Funding; Amgen: Research Funding. Drenou:Alexion: Consultancy, Honoraria. Wagner Ballon:Alexion: Consultancy, Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal