Background: Despite therapeutic advances, AML remains a disease in which the majority of patients relapse after attaining remission. The presence of bone marrow MRD is associated with impending AML relapse. Prediction and prevention of relapse could improve outcomes, but most current MRD tests either require bone marrow aspirate or detect individualized molecular genetic features present in a minority of AML. Furthermore, bone marrow MRD assessments are impractical if performed frequently enough to detect most early relapses. We developed and tested a novel microfluidic chip device (MCD) that can quantitate cell numbers using automated methods and explored its ability to detect low levels of peripheral blood leukemic cells with aberrant immunophenotypes.
Methods: The MCD contains sinusoidal capture channels that were coated with antibodies with specificity towards one of the commonly expressed markers found on immature myeloid cells--CD33, CD34 and CD117 (capture antigens). Initial spiking experiments used fluorescently labeled leukemia cell lines HL60 (CD33+), KG1 (CD34+), Kasumi1 (CD117+) spiked into a 5000 cell/mL suspension. Cell suspensions were passed through MCDs coated with a capture antigen known to be expressed on the tested cell line. These experiments established an efficient capture using a flow rate of 1mL/sec. Then, AML patients with an aberrant immunophenotype were enrolled in a pilot study either at the start of induction chemotherapy or prior to allogeneic stem cell transplant (SCT). For patients receiving induction chemotherapy, whole blood samples were obtained monthly starting at the time of remission assessment, or monthly starting prior to SCT. Buffered whole blood was passed through MCDs coated with a capture antigen known to be expressed on patient myeloblasts. The captured cells were then released, eluted, centrifuged and plated on a glass slide. Plated cell pellets were then labeled with fluorescent antibodies targeting surface proteins known to be aberrantly (either by lineage infidelity or asynchronous expression) expressed on the patients' AML blasts. Automated fluorescence microscopy was used to identify and quantify captured cells with the known aberrant immunophenotype of the AML blasts. Descriptive statistics described serial cell counts in patients maintaining remission and relapsing patients.
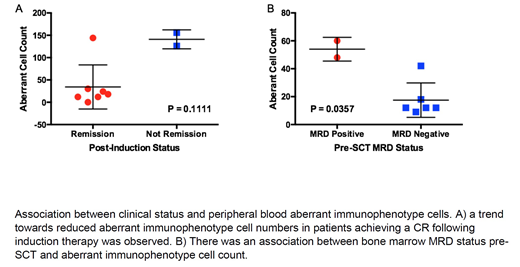
Results: Of 31 patients who have been enrolled in the study to date, 13 had at least 3 post-remission MCD analyses. Of these patients, 6 had either morphologic relapse or persistent/rising marrow MRD. In these patients, there was a trend towards higher initial aberrant immunophenotype cell counts, with mean initial count = 59 (95% CI 1, 108), compared to other patients with mean initial count = 15 (95% CI 5, 25). Of 5 patients who relapsed with MCD data within 1 month prior to relapse, the mean absolute rise prior to relapse above minimum MCD cell count was 54 (95% CI 2, 105), in comparison to non-relapsing patients with mean rise of 9 (95% CI 3, 15). From the initial 16 patients, 10 underwent induction therapy (the other 6 were enrolled prior to SCT). In these 10 patients there was a non-significant association between peripheral blood aberrant immunophenotype cells and remission status following induction. A total of 8 patients underwent allogeneic SCT. Two of these patients had known bone marrow MRD at the time of SCT and had a statistically significant greater number of aberrant immunophenotypic cells pre-SCT (48 and 60) compared to the 6 MRD negative patients (median = 12, range 9, 42).
Conclusions: A novel MCD assay can reliably capture and detect low numbers of AML blasts from peripheral blood using immunofluorescent imaging and automated cell counts to quantify leukemia cells with aberrant immunophenotypes. Because this method uses peripheral blood, frequent sampling is feasible and of minimal risk to patients. An ongoing clinical trial will further explore the associations between MCD-based cell enumeration and clinical endpoints in AML patients that were suggested in the pilot phase of this study. Because the MCD releases trace populations of viable cells, additional experiments, such as primary cell culturing and single cell sequencing, are possible.
Foster:Bellicum Pharmaceuticals, Inc: Research Funding; Daiichi Sankyo: Consultancy; MacroGenics: Research Funding; Celgene: Research Funding. Fedoriw:Alexion Pharmaceuticals: Consultancy, Speakers Bureau. Zeidner:Celgene: Consultancy, Honoraria, Research Funding; AsystBio Laboratories: Consultancy; Merck: Research Funding; Covance: Consultancy; Pfizer: Honoraria; Agios: Honoraria; Daiichi Sankyo: Honoraria; Tolero: Honoraria, Research Funding. Coombs:Covance: Consultancy; Octopharma: Honoraria; Medscape: Honoraria; Cowen & Co.: Consultancy; Loxo: Honoraria; H3 Biomedicine: Honoraria; Dedham Group: Consultancy; Pharmacyclics: Honoraria; Abbvie: Consultancy. Mirkin:BioFluidica: Employment. Zomorrodi:BioFluidica: Employment. Toughiri:BioFluidica: Employment. Bartakova:BioFluidica: Employment. Carson:BioFluidica: Employment. Muller:BioFluidica: Employment.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal