Introduction. The combination of rituximab and lenalidomide (R2) is active in patients with untreated indolent lymphoma. Recent randomized trials (RELEVANCE) have demonstrated similar efficacy when compared to standard chemo-immunotherapy backbones. Long term follow up of patients receiving R2 as well as predictors of long term remission and survival have yet to be published.
Methods. We prospectively evaluated patients with low grade advanced stage FL who received R2 as initial treatment at our institution between 07/2008 and 10/2014. Lenalidomide was given at 20 mg (day 1-21, in a 28 day cycle) for 6 cycles with rituximab monthly. Lenalidomide starting dose was 10 mg if baseline creatinine clearance was < 60 mL/min. Patients with an objective response continued with 10-20 mg of lenalidomide with rituximab for up to 12 more cycles. Response was evaluated according to 2014 Lugano criteria.
Results. One-hundred and one patients were included in the analysis, baseline characteristics are shown in the Table. Median number of provided cycles was 7 (range, 1-20). Median dose of lenalidomide was 20 mg (range, 5-20 mg), and 29 (29%) patients required a dose reduction. Fifty-six (55%) patients experienced grade 3-4 treatment-related toxicities, the most common (> 5%) being neutropenia (39%), skin rash (20%), myalgia (16%) and fatigue (16%). Seven (7%) patients discontinued treatment before completion, after a median time of 4 months (range, 1-10 months): 4 because of toxicity (arterial thrombosis in 2, respiratory failure in 1, and skin rash in 1), and 3 because of progression.
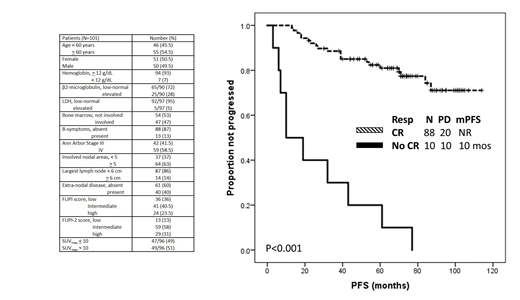
Ninety-eight patients were evaluable for response, while 3 patients discontinued treatment because of toxicity before first response assessment. Overall response rate was 98%, CR rate 90% (both achieved after a median of 6 months [range, 3-22 months]), and CR rate at 30 months (CR30) was 80%. Only female sex associated with a higher CR rate (96% vs 83%, p=0.05), while no baseline characteristic associated with CR30 rate.
After a median follow-up of 88 months (95% confidence interval, 84-92 months), 31 (31%) patients progressed and/or died, 7-year progression-free survival (PFS) was 63%, and 13% of patients had a PFS < 24 months (PFS24). Failure to achieve CR was the only factor associated with significantly decreased PFS (10 months vs not reached, p<0.001) and higher likelihood of PFS24 (46% vs 5%, p<0.001). No association was observed with baseline characteristics, including FLIPI and FLIPI-2 score.
At most recent follow-up, transformation was reported in 3 (3%) patients, after 30, 32 and 42 months, respectively. Two (2%) patients have died, 1 of unrelated comorbid health conditions, 1 of progressive disease, and 7-year overall survival was 98%. Second cancers (excluding transformation) were diagnosed in 8 (8%) patients, after a median of 55 months (range, 3-105 months). These included: breast adenocarcinoma (2), melanoma (2), pancreatic adenocarcinoma (1), esophageal adenocarcinoma (1), and therapy-related acute myeloid leukemia.
Discussion. Long-term follow-up show very favorable outcomes for patients with advanced stage FL receiving R2 as initial treatment, independent of traditional prognostic factors relevant to patients treated with chemoimmunotherapy, including FLIPI and FLIPI-2 score. Combination strategies, aimed at increasing depth of response to R2, may further improve outcomes observed with this regimen.
Nastoupil:Bayer: Honoraria; Genentech, Inc.: Honoraria, Research Funding; Celgene: Honoraria, Research Funding; Gilead: Honoraria; Janssen: Honoraria, Research Funding; Novartis: Honoraria; TG Therapeutics: Honoraria, Research Funding; Spectrum: Honoraria. Westin:Janssen: Other: Advisory Board, Research Funding; Unum: Research Funding; Curis: Other: Advisory Board, Research Funding; 47 Inc: Research Funding; Genentech: Other: Advisory Board, Research Funding; Juno: Other: Advisory Board; Celgene: Other: Advisory Board, Research Funding; MorphoSys: Other: Advisory Board; Novartis: Other: Advisory Board, Research Funding; Kite: Other: Advisory Board, Research Funding. Wang:AstraZeneca: Consultancy, Honoraria, Research Funding, Speakers Bureau; MoreHealth: Consultancy, Equity Ownership; Acerta Pharma: Consultancy, Research Funding; BioInvent: Consultancy, Research Funding; Pharmacyclics: Honoraria, Research Funding; Janssen: Consultancy, Honoraria, Research Funding, Speakers Bureau; Juno Therapeutics: Research Funding; Dava Oncology: Honoraria; Celgene: Honoraria, Research Funding; Aviara: Research Funding; Kite Pharma: Consultancy, Research Funding; Guidepoint Global: Consultancy; VelosBio: Research Funding; Loxo Oncology: Research Funding. Neelapu:Pfizer: Consultancy; Precision Biosciences: Consultancy; Merck: Consultancy, Research Funding; Celgene: Consultancy, Research Funding; Allogene: Consultancy; Novartis: Consultancy; BMS: Research Funding; Kite, a Gilead Company: Consultancy, Research Funding; Cellectis: Research Funding; Acerta: Research Funding; Karus: Research Funding; Poseida: Research Funding; Incyte: Consultancy; Cell Medica: Consultancy; Unum Therapeutics: Consultancy, Research Funding. Fowler:Roche: Membership on an entity's Board of Directors or advisory committees, Research Funding; ABBVIE: Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis Pharmaceuticals Corporation: Consultancy; TG Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding.
lenalidomide and rituximab are not yet FDA-approved as frontline treatment for patients with FL
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal