Background: Therapies involving adoptive transfer of chimeric antigen receptor-modified T-cells (CARTs) targeting CD19-expressing B-cells have shown remarkable efficacy in patients with relapsed/refractory (r/r) diffuse large B-cell lymphoma (DLBCL). We hypothesized, that a higher fraction of more differentiated, senescent and exhausted T-cells affects negatively ex vivo T-cell expansion in the manufacturing phase of CARTs and the in vivo function of CARTs after infusion. Of note, naïve CD4+ T-cells were shown to uniformly express the co-stimulatory receptors CD27 and CD28, while repeated cycles of activation led to their progressive loss (van Leewen et al, J. Immunol, 2004), accompanied by telomere erosion and replicative senescence (van Baarle et al., Immunol. Lett. 2005; Effros RB et al., Dev Comp Immunol 1997). CD28 expression on T-cells within the tumor environment was shown to be a decisive factor for the efficacy of anti-PD-1 therapy (Kamphorst RO et al, Science 2017).
We have therefore analyzed the CD27 and CD28 expression status of CD3+ T-cells from the peripheral blood and apheresis products of adult r/r DLBCL patients at the day of leukapheresis.
Methods: Peripheral blood and apheresis samples of 22 consecutive r/r DLBCL patients scheduled for CART therapy were analyzed by flow cytometry to assess their CD27 and CD28 expression status on CD3+ T-cells. Samples were stained with fluorochrome conjugated antibodies (anti-CD3 PerCP, anti-CD27 FITC, CD28 PE, anti-CD4 FITC, BioLegend) and analyzed using a FACScalibur flow cytometer supported by CELLQUEST software (Becton Dickinson, BD, Palo Alto, CA).
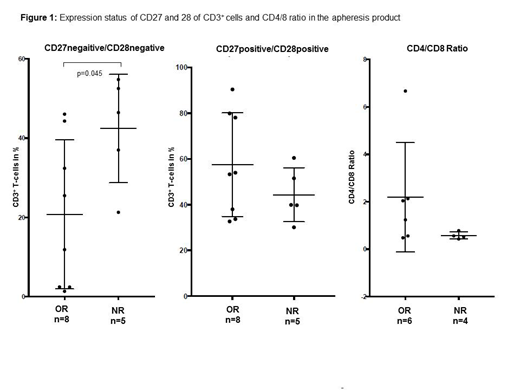
Results: To rule out an apheresis-related bias within cell populations we analyzed peripheral blood and apheresis samples for each patient. No differences in the distribution of CD27-, CD28-, CD27-/CD28- or CD27+/CD28+ T-cells between peripheral blood and apheresis product were detected. Mean CD3+ cell count in blood samples before apheresis was 624±399/µl (range, 75-1853cells/µL) with only about 25% of the patients presenting with CD3+ cell counts within the normal range (690-3320/µL) and 70% of the patients showed an inverse CD4/CD8 ratio (<1.0). We observed a considerable heterogeneity within the T-memory cell compartment. In all samples high percentages of CD27- (39.7±18.1%), CD28- (40.2±19.0%) and CD27-/CD28- (30.7±19.8%) T-cells were seen when compared to healthy controls (CD27-: 15.2±9.9%; CD28-: 18.4±8.9%; CD27-/CD28-: 9.9±8.7%; p<0.05). Patients receiving CARTs were embedded within three different CART-trials (NCT02445248, NCT03630159, NCT03484702). A significantly lower frequency of CD27-/CD28-T-cells (20.8±18.8 vs 42.4±13.7%; p=0.045 Mann Whitney U) was found in responders (n=8) compared to non-responders (n=5; Figure 1a). We also noticed a higher frequency of CD27+/CD28+ T-cells (57.5±22.7% vs 44.3±11.7%; Figure 1b) and a trend towards a normal (>1.0) CD4/CD8 ratio (Figure 1c) in responding patients. Furthermore, we did not observe significant differences in CD27-and CD28- expression in samples derived from patients who died prior to receiving CART therapy (n=6) when compared to patients responding or progressing after CART therapy. Three patients have not been infused yet.
Conclusion: We demonstrate in this small patient cohort that individuals with a lower percentage of more differentiated, senescent or exhausted T-cells are more likely to respond to CART therapy. Our observation underscores the importance of T-cells with normal replicative capacity in the apheresis material for consecutive CART production to achive therapeutic success. Further analysis is needed to determine the effect of cytotoxic pretreatment on the fraction of immunosenescent/exhausted T-cells. However, to confirm our findings additional investigations including the T-cell status of manufactured cells are warranted.
Worel:Sanofi Genzyme, Malinckrodt Therakos: Speakers Bureau; Sanofi Genzyme, Malinckrodt Therakos: Research Funding; Jazz, Sanofi, Celgene, Novartis, Malinckrodt Therakos: Honoraria. Jaeger:Novartis, Roche, Sandoz: Consultancy; AbbVie, Celgene, Gilead, Novartis, Roche, Takeda Millennium: Research Funding; Celgene, Roche, Janssen, Gilead, Novartis, MSD, AbbVie, Sanofi: Membership on an entity's Board of Directors or advisory committees; Amgen, AbbVie, Celgene, Eisai, Gilead, Janssen, Novartis, Roche, Takeda Millennium, MSD, BMS, Sanofi: Honoraria. Hopfinger:Celgene, Gilead, GlaxoSmithKline, Janssen, Novartis, Roche, Takeda,: Honoraria; Gilead: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal