BACKGROUND
FL is the most common indolent non-Hodgkin lymphoma (NHL). While very responsive to therapy, it has been considered incurable. Nonetheless, pts remaining in remission >24 months appear to have a survival comparable to an age-matched population without NHL (Casulo et al, J Clin Oncol, 33:2516, 2015), and some are free of disease for many years and die from unrelated events.
METHODS
Adult pts were accrued from the Lombardi Comprehensive Cancer Center Lymphoma clinic. Pts were required to have histologically confirmed FL or transformed FL that was previously treated resulting in a complete remission, and were required to be free of progression at any time > 24 months following completion of treatment without intervening therapy.
Original diagnostic samples were retrieved and subjected to clonality assessment using Adaptive's next generation sequencing (NGS) MRD assay , a research version of clonoSEQ®; (Adaptive Biotechnologies, Seattle, WA) that leverages multiplex PCR followed by NGS to identify and track rearrangements of IgH, V-J, D-J and IgK/L loci as well as translocations in Bcl1/2 IgH. Lymph node biopsy from time of original diagnosis was assessed to identify trackable clonotypes, which were found in 37/43 patients. Peripheral blood was assayed upon entry onto the study and every 6 months thereafter by the NGS-MRD assay to monitor MRD. Samples are being collected every 6 months during follow-up, and the results are being correlated with clinical outcome.
RESULTS
Of the 60 eligible pts who signed consent 41% were females, with a median age at diagnosis of 56 yrs (21-75) and median age at treatment of 56 years (21-75). Twenty six had received one prior line of treatment (LOT), 4 had 2, 6 had 3, and 1 had 5. The most common immediately prior line of therapy included bendamustine and rituximab (BR, n=16); rituximab, cyclophosphamide, adriamycin, vincristine, prednisone (RCHOP, n=6); double-monoclonal antibody containing regimens(rituximab-galiximab; rituximab-epratuzumab (n=3)), radioimmunotherapy (n=3), and allogeneic bone marrow transplant (n=2). The media follow-up since the start and completion of most recent therapy was 62 months (range 25-183 and 32-193, respectively).
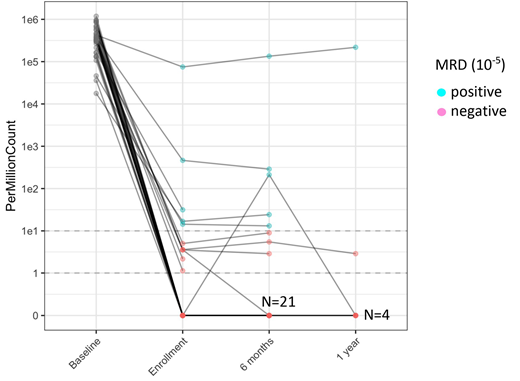
Of the 60 pts for whom original biopsy slides were obtainable, the quality was inadequate to amplify the DNA in 18. In another 5 pts, the sample was polyclonal and a dominant rearrangement could not be identified. In 32 of the 37 pts (86.5%), samples were negative at enrollment to this study at a level of detection of 10-5. By prior LOT, samples were negative in 25 of 26 following 1st line; 1 of 4 following 2nd line; 5 of 6 following 3rd line; and in the one pt after 5th line. In all but 1 pt, the assay has remained negative on subsequent determinations as shown in the spider plot (Fig 1). The 5 positive patients had been followed for a median of 85 (56-118) months. Additional follow-up is underway to determine if positive pts will eventually relapse.
CONCLUSIONS
These data are the first to demonstrate that a high proportion of FL pts in a prolonged clinical remission have undetectable DNA by sensitive next generation sequencing, without evidence of clinical progression, and are potentially cured of their disease.
Yazdy:Bayer: Honoraria, Speakers Bureau; Genentech: Research Funding; Abbvie: Consultancy; Octapharma: Consultancy. Kuhr:Adaptive Biotechnologies: Employment, Other: shareholder. Jacob:Adaptive Biotechnologies: Employment, Other: shareholder. Cheson:Epizyme: Research Funding; TG Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Seattle Genetics: Research Funding; Bristol Myers Squibb: Research Funding; Portola: Research Funding; Kite: Research Funding; Gilead: Research Funding; Genentech: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Pharmacyclics: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Symbios: Equity Ownership, Membership on an entity's Board of Directors or advisory committees; Abbvie: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Trillium: Research Funding; AstraZeneca: Membership on an entity's Board of Directors or advisory committees; Morphosys: Membership on an entity's Board of Directors or advisory committees; Acerta: Consultancy, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal