Background
Renal impairment is a life threatening complication of myeloma with up to 20-25% of patients presenting with renal dysfunction. Outcome is poor as a result of a high early mortality. Around 28% of newly diagnosed myeloma patients with renal failure do not survive beyond 100 days compared with 10% overall. Studies have shown that within weeks of diagnosing myeloma with renal failure, treatment with dexamethasone alone or combined with bortezomib lowers serum free light chain (sFLC) levels by more than 50% in half of patients; achieving lower sFLC levels in this early period is associated with a greater chance of being alive and dialysis free at 100 days.
Methods
OPTIMAL is a randomised, multi-centre phase II trial of newly diagnosed myeloma patients with renal impairment. Renal impairment defined as <30 mls/min were eligible to be included in the trial if they were >18 years old, chronic kidney disease (CKD) stage 4 or 5, not pregnant or risk of pregnancy for child bearing women, or partner of male participants, free of malignancies for >2 years, able to comply with all trial requirements and give fully informed consent. Patients were randomised to receive 4 cycles of either Bortezomib, Bendamustine and Dexamethasone (BBD) or Thalidomide, Bendamustine and Dexamethasone (BTD); all participants received bendamustine and dexamethasone in three week cycles. Treatment period for participants receiving 4 cycles of therapy was 12 weeks. Participants not considered suitable for autologous stem cell transplant (ASCT) could be given a further two cycles of treatment (up to 6 cycles in total) in their respective arms.
The trial was powered to detect 23% differences in the percentage of patients achieving >50% reduction in sFLC between treatment arms, e.g. from 60% to 83%, with 80% power and a 5% 2-sided significance level, recruiting 60 patients in each arm. At the pre-planned interim analysis, the data and safety monitoring committee endorsed the closure of the trial as a 60% difference in sFLC was detected and there was no obvious benefit for the BTD arm. This was also endorsed by the trial steering committee and trial management group.
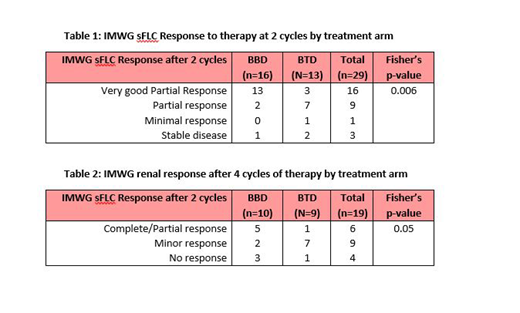
Co-primary endpoints were sFLC response from baseline to week 6 (after receiving two cycles of trial treatment) and renal response according to the modified International Myeloma Working Group (IMWG) criteria after receiving four cycles of trial treatment. Secondary endpoints included haematological responses, toxicity and overall survival.
Results
OPTIMAL recruited 31 patients between March 2015 and March 2019 from seven centres within the UK; 16 on BBD and 15 on BTD. Fifty two per cent of patients were ≤70 years old, 55% male, 35% were CKD stage 4 and 65% were CKD stage 5, 48% had planned autologous-stem cell transplantation, 75% had ECOG performance status 0 or 1, 29% were on dialysis and 90% were ISS stage III. Serum free light chain response was assessed in 29 patients where samples were available at screening and at the end of two cycles of treatment. 81% of patients on BBD achieved vGPR compared to 23% on BTD, Fisher's p=0.006, table 1. Nine patients were on dialysis at the time of screening (6 on BBD and 3 on BTD). Complete or partial renal response was achieved by 2 (50%) of patients on BBD compared to 1 (11%) on BTD, Fisher's p=0.05, table 2. Two patients on BBD arm reported reversibility of dialysis dependency after four cycles of treatment. Two patients not previously on dialysis required dialysis after 4 cycles of BTD. Seven deaths were reported from the total 31 patients (5 (31%) on BBD arm and 2 (13%) on BTD arm). There were 33 reported serious adverse events (SAEs) 14 on BBD and 19 on BTD.
Conclusion
OPTIMAL demonstrated a significant increase in the number of sFLC responders after the first 2 cycles for those patients allocated BBD compared to BTD; this trend continued when assessing renal response after 4 cycles with more patients being dialysis independent after receiving BBD.
Funding: Project funded by NAPP Pharmaceuticals, JANSSEN-Cilag Ltd and Bloodwise (formerly named Leukaemia and Lymphoma Research).
Ramasamy:Oncopeptides and Sanofi: Honoraria, Membership on an entity's Board of Directors or advisory committees; Abbvie: Honoraria, Membership on an entity's Board of Directors or advisory committees; Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; NAPP Pharmaceuticals Ltd.: Research Funding; Janssen-Cilag Ltd.: Research Funding. Drayson:Abingdon Health: Consultancy, Equity Ownership.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal