Introduction: Currently there are four commercially available oral anti-Xa agents namely apixaban, betrixaban, edoxaban and rivaroxaban available for indication specific clinical use. All agents used are at a fixed dosage and their use may be associated with potential hemorrhagic complications. Although factor Xa inhibitory effect is considered to be a surrogate marker for the biologic action of these drugs we have demonstrated that factor Xa inhibitory profile of apixaban, betrixaban, edoxaban, and rivaroxaban does not fully reflect their biologic spectrum (https://doi.org/10.1177/1076029619847524). Furthermore the prothrombin time (PT) and activated partial thromboplatin time (aPTT) methods are of limited sensitivity and are dependant on several other enodogenous factors. Prothrombinase induced clotting time (PiCT), (Pentaharm, Basel, Switzerland) is a sensitive test for the global monitoring of anticoagulant drugs including heparins and parenteral oral anti-Xa and anti-IIa agents. The test is based on the RVV venom activation of endogenous FVa which forms prothrombinase complex with phospholipids (PL), FXa and calcium. The clot-based end point is proportional to the activities of FXa and FIIa. This study is designed to compare the relative responses of PT, aPTT and PiCT test in normal human blood, retrieved plasma and agents supplemented plasma with the oral anti-Xa agents to demonstrate the relative sensitivity of this assay in comparison to the PT and aPTT.
Materials and Methods: Active pharmaceutical ingredient versions of apixaban, betrixaban, edoxaban and rivaroxaban were obtained from commercial sources. All agents were prepared at a stock concentration of 100ug/ml in saline and diluted to a working solution of 10ug/ml. Serial dilutions were prepared in various matrices. Citrated plasma from normal individuals (n=50) was obtained from a commercial source (George King Biomedical, Overland Park, Kansas). Whole blood and plasma samples (n=20) were supplemented at a concentration of 0-1000 ng/ml. Whole blood samples were also centrifuged to obtain retrieved plasma. These samples were analyzed using a single stage PiCT, aPTT and PT. Whole blood and retrieved plasma studies were carried out by using ST4 (Diagnostica Stago). PiCT were measured by using Pefakit PiCT. aPTT measurements were made by using Tcoag, TriniCLOT aPTT (Diagnostica Stago, Paris, France). For PT, HemosIL TM (Instrumentation Laboratory, MA, USA) was used. All results were compiled in individual groups as mean + SD.
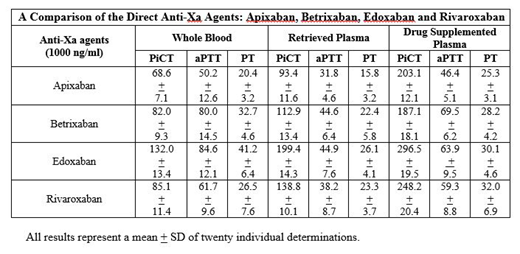
Results: All of these drugs produced a concentration dependent anticoagulant effects in the PiCT, aPTT and PT tests in both the whole blood and plasma systems. PiCT consistently showed much higher sensitivity in comparison to other tests. When compared at 1000ng/ml the anticoagulant effects of these drugs were stronger in plasma systems as shown in the table. PiCT consistently showed higher sensitivity in comparison to PT and aPTT in the whole blood, retrieved plasma and agent supplemented plasma. Edoxaban showed the strongest anticoagulant activity measured by PiCT in comparison to the other agents. Matrix based variations in the PiCT results were observed. Interestingly, the retrieved plasma from whole blood showed weaker anticoagulant effects in comparison to the directly supplemented plasma systems.
Discussion: In comparison to PT and aPTT, the PiCT test was found to be the most sensitive in the three matrices studied. In the whole blood and plasma-based systems PiCT test showed linearity and high sensitivity for all of the four anti-Xa agents. In the PiCT test, consistently drug supplemented plasma showed the highest response in comparison to retrieved plasma and whole blood, suggesting differential binding of these drugs to cells. These results indicate that the PiCT test can be reliably used for the monitoring of anti-Xa agents. PiCT test can also be performed on currently available optical and mechanical instrument used for clotting studies. Owing to rapid turnaround time, high sensitivity, and lower cost PiCT can be used for the routine monitoring of oral anti-Xa agents.
Tafur:Recovery Force: Consultancy; Janssen: Other: Educational Grants, Research Funding; BMS: Research Funding; Idorsia: Research Funding; Daichi Sanyo: Research Funding; Stago: Research Funding; Doasense: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal