Background
Studies from the chemoimmunotherapy (CIT) era and more recently with venetoclax have demonstrated the correlation between minimal residual disease (MRD) response measured by at least four-color flow cytometry (FC), and progression free (PFS) and overall survival (OS) in CLL. Despite high overall (ORR) and complete (CR) response rates observed with fludarabine-based combination CIT, the ability to achieve sustained undetectable MRD (uMRD) remission is lacking for the majority of patients treated with these regimens. We have previously reported on the promising combination of ibrutinib plus FCR (iFCR), which demonstrated a 98.8% ORR, 32.9% CR/CRi with bone marrow (BM) uMRD at EOT, and 77.7% BM-uMRD by flow at EOT (83.5% at best response) [Davids et al, Lancet Haematology, 2019].
Adaptive's next generation sequencing (NGS)-MRD assay targets immunoglobulin receptor sequencing with up to 10E-6 sensitivity for detection of B-cell malignancies. Here we present expanded MRD analysis by standard flow cytometry and the first results assessed using NGS-MRD, focusing on mid-FCR (C3) and 2 month post-FCR (EOT) timepoints.
Methods
iFCR is a multicenter single-arm phase 2 trial at seven sites in the USA. 85 patients aged 65 years or younger with previously untreated CLL were enrolled and treated with iFCR as previously published [Davids, Lancet Haematology, 2019]. Per protocol analyses of MRD in both peripheral blood (PB) and BM by standard four-color FC were performed at local laboratories at C3. Both PB and BM samples were submitted to Adaptive for NGS-MRD evaluation at EOT. Forty-eight patients had paired BM and PB samples with 16 additional PB only samples. NGS-MRD status was evaluated at 10E-5 and 10E-6 levels, and defined as positive if ≥ 1 rearrangement was detected per 100,000 or per million cells, respectively. An indeterminate finding was reported if insufficient cells were assayed, as NGS-MRD testing is limited by the number of cells evaluated, which can often be lower than needed for 10E-6 sensitivity, particularly in PB.
Results
At the C3 restage, the BM-uMRD rate by flow was 47%, with 100% concordance to flow PB-uMRD status in all patients with BM-uMRD. However, 33% (12/36 evaluable) with detectable cells in marrow had PB-uMRD, demonstrating enhanced sensitivity of BM-MRD testing as shown in Table 1. At EOT, BM-uMRD rates rose to 78%, compared with 86% in PB, including 14/24 patients converted from BM-pos/PB-pos to BM-neg/PB-neg and 7/12 BM-pos/PB-neg to BM-neg/PB-neg.
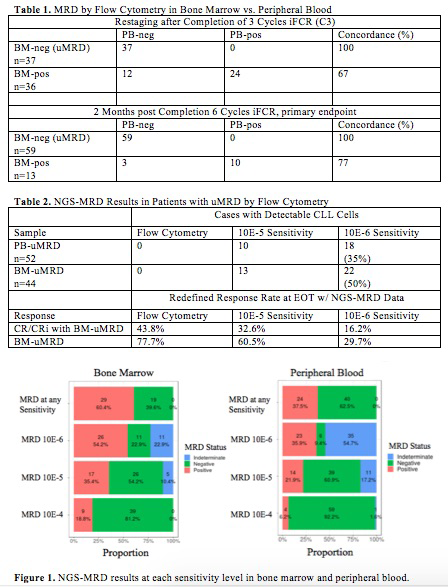
In NGS-MRD analysis from 48 patients with evaluable BM and PB samples at EOT, a larger number of patients were MRD positive in BM (n=21; 43.8%) vs. PB (n=13; 27.1%) (McNemar test: p=0.04). Figure 1 illustrates the improving detection of residual disease in both BM and PB with increasing sensitivity, with greater detection in BM; 54% positive at 10E-6 sensitivity in this cohort, compared with 36% in PB. Evaluation for true negative samples at 10E-6 sensitivity was limited by samples with inadequate cells for evaluation (indeterminate), hence definite uMRD was seen in only 23% BM and 9% PB.
Fifty-two patients with PB-uMRD by FC at EOT had associated PB NGS-MRD results: 10 PB-uMRD by FC were positive at 10E-5 with 8 additional positive at 10E-6 (35% greater than FC). Similar results were observed in BM: of forty-four patients with BM-uMRD by FC at EOT, 13 were positive at 10E-5 with 9 additional positive at 10E-6 by NGS-MRD (50% greater than FC), summarized in Table 2.
When this higher sensitivity BM-uMRD data is used to define overall clinical response at EOT, the CR/CRi with BM-uMRD rate at 10E-5 is 32.6% (14/43), and at 10E-6 is 16.2% (6/37), compared to 43.8% (21/48) using four-color FC. The rate of BM-uMRD would be 60.5% (26/43) at 10E-5 sensitivity and 29.7% (11/37) at 10E-6, with NGS-MRD.
Discussion
This first report of NGS-MRD testing after iFCR demonstrates that 50% of patients with BM-uMRD by flow cytometry have detectable CLL cells at the level of detection of ≥ 1 per million cells. While iFCR has improved upon historical uMRD results by four-color flow cytometry, these findings suggest that CLL cells are still frequently present. Longer follow-up will be required to correlate these minimal levels of residual disease with PFS in this setting. Future studies should incorporate NGS-MRD assessment with larger volume cell sampling to ensure adequate sensitivity and evaluate venetoclax-based regimens.
Brander:Novartis: Consultancy; BeiGene: Research Funding; DTRM Biopharma: Research Funding; AbbVie: Consultancy, Honoraria, Research Funding; Pharmacyclics LLC, an AbbVie Company: Consultancy; MEI: Research Funding; Acerta: Research Funding; Tolero: Research Funding; Teva: Consultancy, Honoraria; TG Therapeutics: Consultancy, Honoraria, Research Funding; Genentech: Consultancy, Honoraria, Research Funding; AstraZeneca: Consultancy, Research Funding. Jacob:Adaptive Biotechnologies: Employment, Other: shareholder. Arnason:Regeneron Pharmaceuticals, Inc.: Consultancy; Celgene/Juno: Consultancy. Abramson:AbbVie Inc, Amgen Inc, Bayer HealthCare Pharmaceuticals, Celgene Corporation, EMD Serono Inc, Genentech, Gilead Sciences Inc, Janssen Biotech Inc, Juno Therapeutics, a Celgene Company, Karyopharm Therapeutics, Kite Pharma Inc, Merck, Novartis, Seattle Gen: Consultancy. Davids:AbbVie, Acerta Pharma, Adaptive, Biotechnologies, Astra-Zeneca, Genentech, Gilead Sciences, Janssen, Pharmacyclics, TG therapeutics: Membership on an entity's Board of Directors or advisory committees; Acerta Pharma, Ascentage Pharma, Genentech, MEI pharma, Pharmacyclics, Surface Oncology, TG Therapeutics, Verastem: Research Funding; AbbVie, Astra-Zeneca, Genentech, Janssen, MEI, Pharmacyclics, Syros Pharmaceuticals, Verastem: Consultancy; Research to Practice: Honoraria. Brown:Novartis: Consultancy; Sunesis: Consultancy; Acerta Pharma: Consultancy; AstraZeneca: Consultancy; BeiGene: Consultancy; Catapult Therapeutics: Consultancy; Dynamo Therapeutics: Consultancy; Genentech/Roche: Consultancy; Gilead: Consultancy, Research Funding; Juno/Celgene: Consultancy; Pfizer: Consultancy; Loxo: Consultancy, Research Funding; Invectys: Other: Data safety monitoring board; Octapharma: Consultancy; Kite, a Gilead Company: Consultancy, Research Funding; AbbVie: Consultancy; Morphosys: Other: Data safety monitoring board; Pharmacyclics: Consultancy; Teva: Honoraria; TG Therapeutics: Consultancy; Verastem: Consultancy, Research Funding; Sun Pharmaceuticals: Research Funding; Janssen: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal