Introduction: The prognosis of patients with multiple myeloma (MM) who are resistant to proteasome inhibitors (PIs) and immunomodulatory drugs (IMiDs) is extremely poor. Even daratumumab and BCMA CAR-T cell therapies appear to provide only temporary benefit before patients eventually succumb to their disease. We show that protein translation is upregulated in MM cells. We also demonstrate that translational inhibition with omacetaxine (Oma) is specifically cytotoxic in a manner that is preserved in MM cells from PI/IMiD resistant patients. Furthermore, protein translation inhibition is synergistic with IMiDs, even in IMiD refractory patients. We hypothesize these findings stem from the origin of MM cells as professional antibody secretory cells, with highly active protein translation fundamental to their biology. Here, we show these findings and provide support to test the translation inhibitor omacetaxine with a clinical trial in multiple myeloma patients.
Methods: Primary MM cells in mononuclear cell (MNC) cultures were assayed ex vivo for Oma effect at 48-hrs by high-throughput flow cytometry using our myeloma drug sensitivity testing (My-DST) platform. Protein synthesis rates were measured via flow cytometry using the Cayman OP-puromycin assay. Initial drug combination matrices were performed in MM cell lines via MTS assay after 96-hr incubation. Cell line xenograft study of the Oma and IMiD combination in vivo were performed using an orthometastatic MM model with MM1.S luciferase-expressing cells injected IV in NSG mice. Vehicle control, Oma, pomalidomide (Pom) and the combination were dosed 5 days/week by IP injection beginning on day 30. Disease burden was monitored with imaging of luciferase bioluminescence.
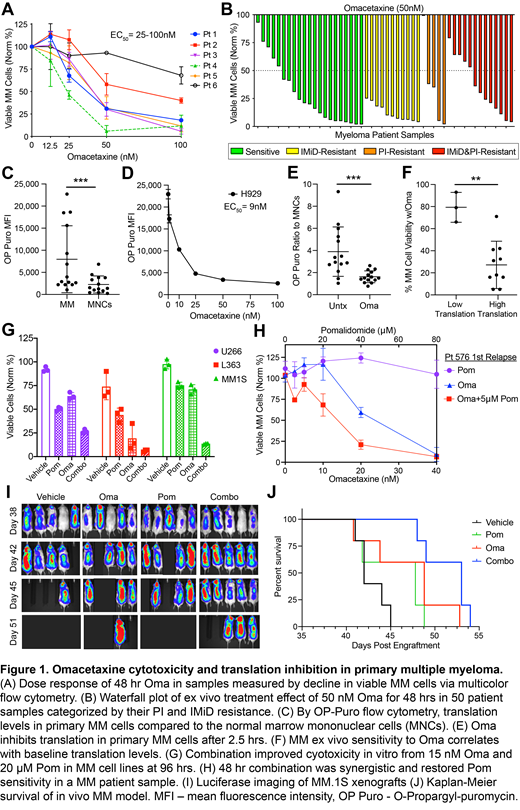
Results: Oma had specific cytotoxicity across 5 MM cell lines (EC50 = 15-35 nM, data not shown). Oma also showed MM cell specific cytotoxicity in patient samples with EC50 ranging from 25-100 nM (Fig 1A). Ex vivo treatment with 50 nM Oma for 48 hrs caused >50% reduction in viable MM cells in 39/50 (78%) patient samples. Importantly, 50 nM Oma retained activity in PI/IMiD-refractory patients compared to sensitive patients (64.2% vs 71.6% mean decrease in MM cell viability, respectively, P = 0.44, Fig 1B). Primary MM cells exhibited significantly higher levels of baseline protein translation than other bone marrow MNCs with mean 3.9-fold increase (n = 14, P = <0.001) (Fig 1C). Oma treatment inhibited protein translation in a dose dependent manner after 2.5 hrs in H929 cells with EC50 9 nM (Fig 1D), and translation in the primary MM cells was significantly reduced after 2.5 hrs of 50 nM Oma treatment to near MNC levels (Fig 1E). Using a cutoff of 2.5-fold higher baseline MM translation compared to MNCs, "High Translation" was associated with sensitivity to Oma, and "Low Translation" was associated with relative resistance (P = 0.0018) (Fig 1F). Provocatively, Oma demonstrated synergy in combination with Pom (5.52 ZIP synergy δ-score) in the MM1.S cell line (Fig 1G), but was antagonistic with PIs (data not shown). Notably, in an IMiD-resistant relapsed patient sample, the combination of Oma and Pom was even more synergistic and re-sensitized the MM cells to the IMiD (22.68 δ-score, Fig 1H). In a MM1.S luciferase xenograft model of advanced MM, Oma treatment significantly extended survival compared to vehicle (HR = 6.67, P = 0.021), Pom treatment led to a trend towards extended survival (HR = 2.78, P = 0.16), and Oma in combination with Pom extended median survival the most (HR = 16.98, P = 0.002) (Fig 1I-J).
Conclusion: Protein translation inhibitors represent a potential new drug class for clinical myeloma treatment. We have shown that the protein synthesis inhibitor omacetaxine possesses a potent and specific killing effect against MM cells. Importantly, this anti-myeloma cytotoxicity was equally active in biopsy samples obtained from patients at diagnosis and patients with disease advanced to the late stage of PI/IMiD resistance. The pretreatment level of MM cell protein translation may serve as a biomarker for clinical response. In further exploration of the optimized treatment approach for clinical trials, we found synergy between omacetaxine and pomalidomide in vitro, in vivo and using patient samples ex vivo. A phase I clinical trial of omacetaxine single agent and in combination with pomalidomide is currently in development at the University of Colorado.
Mark:Janssen: Honoraria; Takeda: Honoraria; Amgen: Honoraria; Celgene: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal