BACKGROUND: Regulatory T cells (Tregs) are non-redundant mediators of immunity and tolerance. Adoptive transfer of CD4+Foxp3+ Tregs has emerged as a promising therapy for graft-versus-host disease (GVHD) following allogeneic HSCT (aHSCT), solid organ transplantation and autoimmune diseases (Hanash AM, Blood 2005; Copsel S, Wolf D, Haematologica 2019; Marek-Trzonkowska N, Diabetes Care 2012; Tang Q, J Mol Cell Biol 2012). Our prior work was the first to demonstrate a two-pathway in vivo strategy targeting TNFRSF25 (with TL1A-Ig fusion protein) and CD25 (with low dose IL-2, IL-2LD) receptors can elicit a rapid and strong increase in Treg numbers and function (Wolf D, BBMT 2017). In fact, very low numbers of these in vivo expanded donor Tregs exhibited effective GVHD suppression in recipients following aHSCT (Copsel S, BBMT 2018). Based on these findings and the success of IL-2LD in pre-clinical and clinical studies, we asked: are there phenotypic/functional differences between Tregs expanded via IL-2LD, TL1A-Ig or their combined application (TL1A-Ig+IL-2LD)?
METHODS: Mice were administered IL-2LD, TL1A-Ig or TL1A-Ig+IL-2LD over 6 days. Splenic and lymph node (LN) Treg phenotype was determined by flow cytometry. Treg functionality was assessed with sorted populations using an in vivo MHC-mismatched aHSCT.
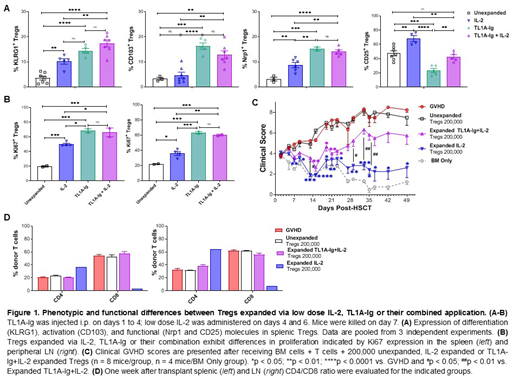
RESULTS: Treatment of C57BL/6-FoxP3RFP mice with TL1A-Ig+IL-2 LD versus IL-2LD only treatment resulted in significantly higher levels of activation/differentiation and functional markers on Tregs including KLRG1, CD103, Nrp1, ICOS (Fig. 1A). Ki67 expression was higher in two-pathway versus IL-2LD stimulation alone (Fig.1B). These data suggested a key role for TNFRSF25 stimulation. Notably, Treg stimulation with TL1A-Ig alone drove the above phenotype indicating a pathway difference between the TNFRSF25 and IL-2 receptors. This difference was further apparent as high affinity IL-2 receptor (CD25) expression was reduced after TL1A-Ig +/- IL-2LD -mediated expansion compared with IL-2 LD alone stimulated Tregs (Fig. 1A). Results were corroborated using a second independent mouse strain, BALB/c, following use of these protocols. To begin addressing if the observed phenotypic differences between CD25 vs. TNFRSF25 + CD25 expanded Tregs could be related to a more potent Treg in vivo suppressive activity, an initial MHC-mismatched aHSCT (donor/recipient = C57BL/6-BALB/c) was performed. We employed 200,000 sorted Tregs (>98% purity by CD4+FoxP3+ selection from C57BL/6-FoxP3RFP reporter mice) from donor unexpanded, IL-2LD, or TL1A-Ig+IL-2LD treated mice combined with 1.0 x106 T cells. As anticipated, transfer 200,000 TL1A-Ig+IL-2LD stimulated Tregs ameliorated acute GVHD (Fig. 1C). Remarkably, lower GVHD clinical scores were obtained using the same number of IL-2LD only expanded Tregs compared with TL1A-Ig+IL-2LD stimulated Tregs (Fig. 1C). Moreover, early post-transplant, higher LN and splenic CD4/CD8 ratios were detected in aHSCT recipients treated with IL-2LD expanded Tregs vs. TL1A-Ig+IL-2LD (Fig. 1D).
CONCLUSION: Our donor TL1A-Ig+IL-2LD Treg expansion protocol promotes a more activated/differentiated and proliferative phenotype versus IL-2LD stimulation alone. This finding may have accounted for their initial effectiveness - but less efficient long-term GVHD amelioration compared to IL-2LD only stimulated Tregs. Multiple variables are associated with the application of Tregs for therapy including numbers, persistence, and suppressive capacity. Our findings suggest a rationale that one-pathway and / or two-pathway stimulated Tregs may be beneficial for use in aHSCT recipients dependent on whether there is a perceived need for prolonged Treg presence and the stage of GVHD.
Levy:Heat Biologics: Consultancy, Equity Ownership, Membership on an entity's Board of Directors or advisory committees, Research Funding; Pelican Therapeutics: Consultancy, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal