Background:
Early data from anti-CD19 chimeric antigen receptor (CAR) T-cell trials suggest that concurrent infection can lead to poor outcomes. 1,2 The relationship between CAR T-cell mediated inflammatory responses and infections is not well-established. With CAR T-cell therapies more readily available, practitioners must identify which patient, disease and CAR T-cell specific parameters are associated with an increased risk of infection to optimize outcomes. We provide a comprehensive analysis of infection risk within the first 30 days after CAR T-cell infusion across multiple types of CAR T-cell trials targeting distinct antigens.
Methods:
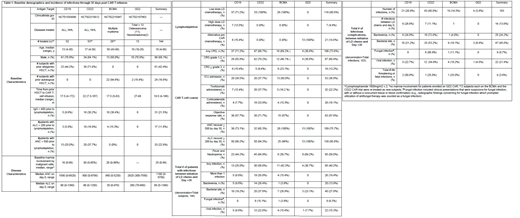
This was a single-center, retrospective study conducted at the National Cancer Institute analyzing infectious complications in subjects who underwent CAR T-cell therapy on one of four phase I clinical trials, targeting CD19, CD22, B-cell maturation antigen (BCMA) or disialoganglioside (GD2) from 2012 though 2018. Baseline characteristics are in Table 1. The primary objective was to establish the incidence of infections between initiation of lymphodepleting (LD) chemotherapy through day 30 after CAR T-cell infusion. The secondary objective was to identify risk factors for infection. Patients were censored at relapse and/or initiation of alternative therapy, including treatment for relapse or consolidative hematopoietic stem cell transplantation (HSCT). Univariate screening methods were used to identify parameters associated with increased risk of infection
Results:
Amongst 144 subjects, 52 (36.1%) received anti-CD19 CAR T-cells (CAR-T); 53 (36.8%) received anti-CD22 CAR-T, 26 (18.1%) received anti-BCMA CAR-T; and 13 (9%) received anti-GD2 CAR-T. The median age was 18 years (range: 4 to 66). Sixty-one (42.4%) had undergone at least one prior allogeneic hematopoietic stem cell transplant (HSCT) and 24 (16.7%) had at least one prior autologous HSCT. Sixty-eight (47.2%) had a history of a recent infection within 100 days of initiation of LD chemotherapy, of which 17 were a major chronic infection and 9 were considered severe.
Fifty-eight (40.3%) subjects experienced a total of 103 infections from initiation of LD chemotherapy through day 30 post CAR T-cell infusion, with the median time to first infection being 6 days post-CAR infusion. Twenty-eight (19.4%) subjects had more than 1 infection; 20 (13.9%) subjects had C. difficile infection. The 103 infections consisted of 47 (45.6%) distinct episodes of focal bacterial infections (e.g., sinusitis, pneumonia, urinary tract infection), 25 (24.2%) episodes of bacteremia, 22 (21.4%) viral infections, and 9 (8.7%) invasive fungal infections. Fourteen infections (13.6%) occurred between initiation of LD chemotherapy and day 0 (prior to CAR T-cell infusion). Fever and neutropenia, without a source, was documented as a distinct entity in 85 patients (59.0%). Using univariate statistical screening methods, we identified 4 parameters that were individually associated with an increased risk of infection: increasing number of prior therapies (p=0.0034), prior history of recent infection within the past 100 days (p=0.0064), age > 18 (p =0.028), or enrollment on the CD22 CAR trial (p=0.0028). Eliminating the trial, since that is not generalizable, and combining these into a multivariable logistic regression model identified age > 18, prior history of infection and prior lines of therapies jointly associated with an increased risk of infection. Using these parameters, a predictive model was developed, which, when applied to the data set used to develop the model, can correctly predict 69 of 86 patients without an infection (80.3%; CI: 70.3-88.0%) as well as 32 of 58 patients with an infection (55.2%; 95% CI: 41.5- 68.3%).
Conclusion:
In this retrospective analysis, we provide the largest experience to date analyzing infection in the setting of CAR T-cell therapy across multiple CAR constructs. Our study demonstrates that adult age, prior history of infection, increased number of prior therapies and enrollment on a particular CAR T-cell trial, in this case the CD22 CAR-T trial, may lead to a higher risk of infection than in those without these risk factors. Further investigations are underway to evaluate clinical outcomes with infection which occur in the peri CAR T-cell setting and potential strategies to minimize infection risk. Generalizability of this model will be tested in an independent data set.
Lee:Kite, a Gilead Company: Research Funding; Harpoon Therapeutics: Consultancy; Juno Therapeutics: Other: External Advisory Board; ACI Clinical on behalf of Celgene:: Other: Independent Central Quality Review Committee. Mackall:Obsidian: Research Funding; Lyell: Consultancy, Equity Ownership, Other: Founder, Research Funding; Nektar: Other: Scientific Advisory Board; PACT: Other: Scientific Advisory Board; Bryologyx: Other: Scientific Advisory Board; Vor: Other: Scientific Advisory Board; Roche: Other: Scientific Advisory Board; Adaptimmune LLC: Other: Scientific Advisory Board; Glaxo-Smith-Kline: Other: Scientific Advisory Board; Allogene: Equity Ownership, Membership on an entity's Board of Directors or advisory committees; Apricity Health: Equity Ownership, Membership on an entity's Board of Directors or advisory committees; Unum Therapeutics: Equity Ownership, Membership on an entity's Board of Directors or advisory committees. Kochenderfer:Kite and Celgene: Research Funding; Bluebird and CRISPR Therapeutics: Other: received royalties on licensing of his inventions.
Conditioning chemotherapy for CAR T-cell therapy; this is a retrospective study that used different lymphodepletion regimens.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal