Background: Deep and durable hematologic remissions following RA-ASCT are associated with improved organ function and extended overall survival (OS) in AL amyloidosis. Achieving at least a very good partial response (VGPR) defined by a dFLC <4mg/dL is an accepted goal of therapy based on favorable outcomes, including improved renal survival (REF: Palladini JCO 2012, Palladini Blood 2014). Recently more profound clonal suppression as indicated by no evidence of minimal residual plasma cell disease (MRD) in bone marrow (BM) (Muchtar Blood 2017) and achieving dFLC <1mg/dL (Manwani Blood 2018) have shown additional benefit. While depth of hematologic response by standard criteria are important, this study assessed additional factors that influence renal response and time to renal response.
Methods: All patients (pts) with AL and renal involvement (biopsy proven renal tissue diagnosis and/or 24hr proteinuria >500mg/day) undergoing RA-ASCT at Memorial Sloan Kettering Cancer Center between January 1, 2007 to December 31, 2016 were included. Pts with follow up less than 12 months post RA-ASCT, hemodialysis prior to RA-ASCT and Waldenstrom macroglobulinemia were excluded. Melphalan dose was assigned based on age, cardiac involvement and renal compromise (Landau Leukemia 2013). Hematologic response was assessed at 3 and 12 months (mos) post RA-ASCT (Palladini JCO 2012) and those with less than complete response (CR) were offered consolidation therapy with bortezomib and dexamethasone (BD). All pts underwent serial organ function assessment (Palladini Blood 2014). Logistic regression models were used to assess association with renal response by 12 mos. Covariates for adjustment in multivariate models were chosen based on univariate analyses and clinical relevance.
Results: Sixty-four patients with renal AL meeting the inclusion criteria were identified; 3 pts died within a year post RA-ASCT were excluded. Median age (range) was 61 years (44-73), M:F 49%:51%, white 90% and 34% had cardiac involvement. Median (IQR) 24 hr proteinuria pre RA-ASCT was 5014 mg/day (2632-7514) and eGFR 68 ml/min/1.73 m2 (44-91). Renal amyloid stage I:II:III was 33%:52%:15%. Mayo cardiac stage (2004) I:II:III was 28%:61%:11% and revised Mayo stage (2012) I:II:III:IV was 13%:57%:21%:8%. Median BM plasma cells pre RA-ASCT was 9% (IQR 2-14%). 46% pts received treatment prior to ASCT. Melphalan dose (mg/m2) 200:140:100 was 44%:43%:11%. 46% pts received BD consolidation.
Hematologic response at 3 mos post RA-ASCT was CR 44%, VGPR 29%, partial response (PR) 20% and stable disease (SD) 7%. MRD in BM by 10-color flow cytometry was assessed in 33 pts and 13 (39%) were MRD negative. dFLC <1mg/dL was achieved in 63% of pts. Renal response by 12 mos following RA-ASCT was achieved in 32 pts (53%). Median (IQR) time to renal response in these pts was 5.8 mos (5.1 - 11.3). Amongst renal responders, 50% were in CR, 53% had MRD negative BM (of 15 pts) and 78% with dFLC <1mg/dL early post RA-ASCT. In pts who achieved dFLC <1mg/dL early post RA-ASCT, 66% had renal response.
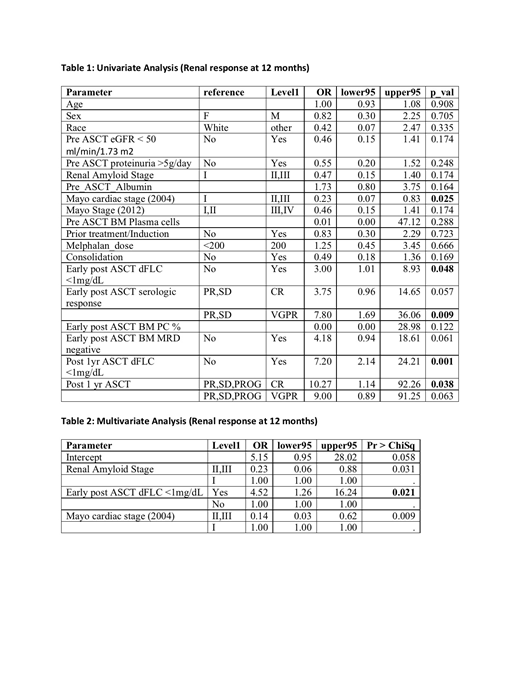
By univariate analysis (Table 1) OR (95% CI) Mayo cardiac Stage (2004) II and III 0.23 (0.07-0.83, p=0.025), early post RA-ASCT dFLC <1mg/dL 3.00 ( 1.01-8.93, p=0.048), VGPR early post RA-ASCT 7.80 (1.69-36.06, p=0.009), dFLC <1mg/dL at 12 mos 7.20 (2.14-24.21, p=0.001) and CR at 12 mos 10.27 (1.14-92.26, p=0.038) were significantly associated with renal response. Neither renal stage, Mayo stage (2012), MRD negativity, melphalan dose nor consolidation was associated with renal response. By multivariate analysis (Table 2), early post RA-ASCT dFLC <1mg/dL continued to be the most significant factor predicting renal response, OR (95% CI) 4.52 (1.26-16.24, p=0.021), when adjusted for renal amyloid stage and Mayo cardiac stage (2004).
Conclusion: In this single center study, we report that RA-ASCT results in renal response in more than half (53%) of the patients at 1 year. Achieving dFLC <1mg/dL early post ASCT is significantly associated with renal response. Renal response is independent of baseline proteinuria and BM plasma cells or MRD status post ASCT. Our study supports that pathologic entity in organ damage is not the plasma cells but rather light chains. Further studies using dFLC <1mg/dL should be evaluated in organ response. Mass spectrometric light chain monitoring may even be more sensitive and could potentially serve as a non-invasive way to measure disease burden.
Shah:Janssen: Research Funding; Amgen: Research Funding. Hassoun:Janssen: Research Funding; Celgene: Research Funding; Novartis: Consultancy. Giralt:Celgene: Consultancy, Research Funding; Takeda: Consultancy; Sanofi: Consultancy, Research Funding; Amgen: Consultancy, Research Funding. Landau:Pfizer: Membership on an entity's Board of Directors or advisory committees; Prothena: Membership on an entity's Board of Directors or advisory committees; Caelum: Membership on an entity's Board of Directors or advisory committees; Amgen: Research Funding; Takeda: Membership on an entity's Board of Directors or advisory committees, Research Funding; Karyopharm: Consultancy, Honoraria; Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal