Bone complications are the most dramatic and life-impairing outcomes of type 1 Gaucher disease (GD1), a common lysosomal storage disorder. Bone marrow infiltration by Gaucher cells substantially decreases the bone marrow fat fraction (FF), and the extent of this reduction correlates with the overall severity of skeletal manifestations in this disorder. Previous studies have demonstrated that the degree of infiltration can best be estimated by magnetic imaging resonance (MRI)-based quantitative chemical shift imaging (QCSI) and that the fat fraction (FF) score so derived can predict the risk of clinically important bone events. The aim of this study was to evaluate bone marrow involvement in GD1 patients who had received enzyme replacement therapy (ERT) for at least 5 years.
Methods: Patients from SZMC Gaucher unit, ≥ 18 years, who were treated with ERT for ≥ 5 years, with a stable dose in the previous 6 months, were recruited. Patients taking another experimental drug, with past exposure to taliglucerase-alfa, presence of any medical, emotional, behavioral or psychological condition were excluded. Energy x-ray absorptiometry (DEXA) was performed at SZMC and the QSCI was performed at the Academic Medical Center in Amsterdam, Netherlands as previously described [Mass et al, Am J Radiol 2002:179:961-965]. A QCSI score of <0.30 was indicative of bone at risk. This investigator initiated clinical trial was approved IRB at SZMC and AMC and sponsored by Pfizer. Study number registration- MOH-2017-04-000351.
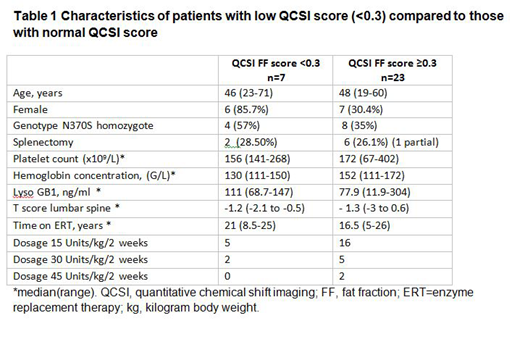
Results: Thirty patients (13 females) at a median (range) age of 46 (19-71) years consented to participate in this study and to perform the QCSI test. GBA mutations of study patients included N370S homozygote (n=12), N370S compound heterozygote (n=17), and T431 homozygote (n=1). The median (range) duration of ERT was 18 (5-26) years. Thirteen patients were receiving imiglucerase as the primary ERT [median (range) duration, 19 (9-26) years], five patients were receiving velaglucerase alfa [median (range) duration, 11 ( 5-12) years], and 12 patients converted from imiglucerase to velaglucerase alfa [median (range) duration, 7 (6-10) years]. The majority of patients received low-dose regimen, i.e. 15 Units/kg/2 weeks (Table 1). The median (range) T score for lumbar spine from DEXA scans, available for 26 of 30 patients, was −1.3 (−2.8-0.0). The median (range) QCSI score was 0.42 (0.24-0.66). Seven patients, 23% (95% confidence interval 10%-42%), had abnormal QCSI FF scores (<0.30). Abnormal QSCI score was more common in female compared to male (Table 1) (p=0.025). Only one of these was menopausal. No differences were found in age, gender, genotype, history of splenectomy, duration and type of ERT and GD-related parameters between those with QSCI score of bone at risk to those with normal score (Table 1).
In summary, these findings demonstrate that, despite prolonged treatment with imiglucerase and/or velalgucerase alfa, 23% of patients still had QCSI scores indicative of an inadequate response in bones. Nevertheless, most patients with prolonged low-dose ERT maintain a normal QCSI, indicative of a positive bone status. The higher prevalence of women in the cohort with low FF is not related to menopausal phase and remains unexplained. As no other patient-related nor GD-related parameter predicted abnormal bone marrow infiltration, a more widely available, quantitative measure of bone marrow infiltration is required for the assessment of response in bones to ERT for patients with GD1. The second phase of this study will evaluate the impact of a switch to a third ERT in those patients from this study with QCSI scores of <0.30. These patients will be offered treatment with taliglucerase alfa at equivalent doses and subsequent reassessment of any impact on clinical symptoms and QCSI scores evaluated.
Revel-Vilk:Takeda: Honoraria, Other: Travel, Research Funding; Prevail therapeutics: Honoraria, Other: Travel, Research Funding; Pfizer: Honoraria, Other: Travel, Research Funding; Sanofi: Honoraria, Other: Travel, Research Funding. Szer:Alexion: Honoraria, Other: Travel, Research Funding; Amgen: Honoraria, Other: Travel, Research Funding; Celgene: Honoraria, Other: Travel, Research Funding; MSD: Honoraria, Other: Travel, Research Funding; Novartis: Honoraria, Other: Travel, Research Funding; Pfizer: Honoraria, Other: Travel, Research Funding; Takeda: Honoraria, Other: Travel, Research Funding; Sanofi: Honoraria, Other: Travel, Research Funding; Prevail Therapeutics: Honoraria, Other: Travel, Research Funding. Zimran:Centogene: Other: research grant; Shire: Consultancy, Honoraria, Research Funding; TAKEDA: Honoraria; Pfize: Honoraria, Research Funding; Bio-events: Honoraria; Targeted Cell Therapies: Consultancy; Prevail Therapeutics: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal