Background
Analysis of SEER database shows that the median age of acute myeloid leukemia (AML) patients at diagnosis is 68 years, with ~1/3 of patients older than 75 years. Although acute lymphoblastic leukemia (ALL) is mainly a disease of the younger population, ~12% of patients are older than 65 years old at diagnosis.
Elderly patients suffer from increased toxicity compared to younger patients translating into a need for reduction in chemotherapy dose intensity as well as a higher rate of early death.
Elderly patients, 70 years of age and older, receiving intensive chemotherapy have an extremely poor outcome with reported median overall survival (OS) of approximately 4 months in ALL and 6-12 months in AML.
Within the last couple of years a flurry of targeted therapies has been introduced into clinical practice and new treatment opportunities for older patients have become available.
We sought to investigate the pattern of targeted therapy utilization and clinical trial enrollment in the older patients with acute leukemia in our center.
Methods
A single center retrospective analysis of patients aged ≥ 70 years with newly diagnosed acute leukemia treated at a the hemato-oncology admission unit in a large tertiary center between 4/2011 and 4/2019.
Results
52 patients [AML (n=44 including 3 acute promyelocytic leukemia), ALL (n=5), mixed phenotype acute leukemia (n=2) and blastic plasmacytoid dendritic cell neoplasm (n=1)] were included in the analysis.
44% of the entire cohort received novel approaches with 26.9% of patients enrolled in clinical trials and 30.7% receiving targeted therapy in the off-trial setting (in all lines of therapies).
Due to the small cohort of non-AML patients, we present herein in detail the results of the non-APL AML patients only (Table 1).
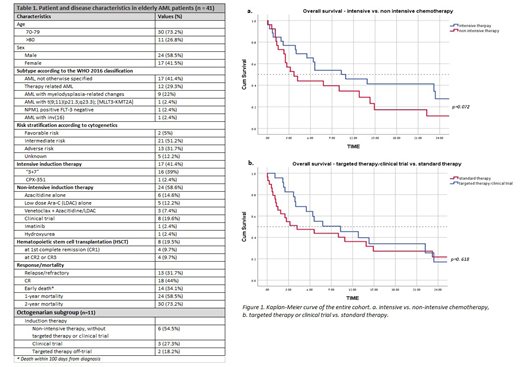
Most of the AML patients had high-risk features such as therapy related AML (n=12), AML with myelodysplastic-related changes (n=9). Adverse risk cytogenetics was observed in 13 patients.
17 patients (41%) received intensive chemotherapy, mainly the "3+7" induction regimen with daunorubicin 60mg/m2/day. 58.6% were considered ineligible for intensive chemotherapy due to age and comorbidities, third of those patients were enrolled into clinical trials, and two-thirds received either Azacitidine alone, LDAC alone, or in combination with venetoclax.
The most used targeted agent was venetoclax (combined with either azacytidine or LDAC) which was given to 12 patients (all lines of therapies) achieving 1-year OS of 50%.
The complete remission (CR) rate for patients receiving intensive chemotherapy was 41% and the 1-year OS was 47.1% with a median OS of 10.3 months compared to 37.5% CR and a median OS of 3.7 months for patients receiving non-intensive therapy (95% CI, 1.4-6, p=0.072, Figure 1a). Patients who received either targeted therapy or were enrolled upfront into a clinical trial had a 1-year OS of 44.4%.
Four patients, median age of 73.5 years (70.7-73.7 years), underwent hematopoietic stem-cell transplantation in first remission , receiving non-myeloablative conditioning, with median OS of 75% at 1 year of follow-up.
In a subgroup analysis of octogenerians (≥ 80 years old), all 11 patients were diagnosed with AML, and 63% of them had either therapy-related AML or AML with myelodysplastic-related changes. All received non-intensive therapy with 36% receiving either targeted therapy or enrolled into a clinical trial. Interestingly, although the1-year OS of the octogenerians was 36%, those on a clinical trial or receiving targeted therapy had 1-year OS of 91%.
For the entire cohort (including non-AML patients), patients who were either enrolled into a clinical trial or received targeted therapy had 1-year OS of 47.8% with a median OS of 10.3 months compared to 37.9% and median OS of 4.2 months 1-year OS for those who did not receive targeted therapy or were not enrolled into a clinical trial (95% CI, 1.8-18.8, p=0.618, Figure 1b).
Conclusions
Patients over 70 years of age with acute leukemia seem to benefit from the administration of targeted therapy and/or enrollment into clinical trials compared to standard approaches. This holds true also for octogenerians. If this is not feasible, a selected group of carefully chosen patients might benefit from intensive therapy as well.
Wolach:Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Speaker; AbbVie: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Speaker.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal