Introduction: Chronic lymphocytic leukemia (CLL) is the most common type of leukemia seen in adults. Ibrutinib is an orally administered irreversible inhibitor of Bruton tyrosine kinase. Ibrutinib monotherapy has been found effective in many CLL studies. This study aimed at investigating the real world experiences of patients with CLL who have been treated with ibrutinib in Turkey.
Materials and Methods: This retrospective multicenter study included 136 patients (91 male) from 33 sites who received at least 1 cycle of ibrutinib since February 2013.
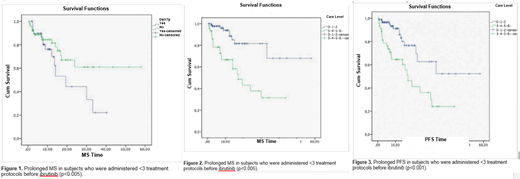
Results: The median age was 64. The median follow up time from the initial diagnosis was 69 months (range: 9-296). An average of 2 treatment protocols had been given (range: 0-7) before ibrutinib and the median time elapsed from the last treatment to the date of ibrutinib administration was 6 months (range: 0-120 months). Before ibrutinib, 51.5% of the patients had a comorbidity, 21.3% a bulky disease and 52.2% a B symptom. Of the patients, 87.9% had ECOG <3, 23.8% RAI stage <III, 38.7% 17p deletion, 7.5% 11q deletion and 1.9% p53 mutation. The median follow up time from the initiation of ibrutinib was 8.8 months (range: 0.4-58 months). The median number of ibrutinib cycles was 8 (range: 1-58). The rates of response to ibrutinib were 26% complete remission (CR), 40.7% partial remission (PR), 9.8% stable disease (SD), and 23.6% progressive disease (PD). The overall rates of response (ORR) were (79.3%-41.5%, p<0.005) in those who received <3 treatments before ibrutinib, (75%-50%, p=0.006) in those with ECOG <3 and more (88.9%-57%, p=0.002) in those with RAI stage <III. Presence of 17p deletion (p=0.409), an accompanying disease (p=0.197) or a bulky disease (p=0.751) did not affect the ORR. The dose of ibrutinib was reduced in 17% and delayed in 26.5% of the patients. The major reason for these was the side effects that developed in 55.2% of the cases. The most common adverse events were GI-related (n=37: Grade 1-2 diarrhea in 17 subjects, grade 1-2 nausea in 6 subjects, grade 1 elevated liver enzymes in 4 subjects) and hematologic-lymphatic system toxicities (n=49: Anemia in 19 patients [grade 1-2 in 14], neutropenia in 18 patients [grade 1-2 in 7], thrombocytopenia in 10 patients [grade 1-2 in 5]). Pneumonia developed in 19 subjects (grade 1-2 in 7). The proportion of the patients who survived throughout the study was 78.7%. The most common cause of death was sepsis. The median progression-free survival (PFS) was 30 months and the median survival (MS) rate could not be reached. The 12-month MS rate was 84.6% and the 36-month MS rate 51.7%. The PFS and MS times were found longer in those who had been administered <3 treatment protocols before ibrutinib (p=0.001 for PFS and p<0.005 for MS), those with ECOG <3 (p=0.011 for PFS and p=0.001 for MS), and those with RAI stage <III (p=0.001 for PFS and p=0.002 for MS). Although the PFS and MS times decreased in the presence of 17p deletion, it was not statistically significant (p=0.224 and p=0.123, respectively).
Conclusion: These real world experiences have shown that the ibrutinib monotherapy with its tolerable side effects is an effective treatment option regardless of the poor prognostic gene mutations in patients with CLL
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal