Original versus generic lenalidomide in patients with relapsed multipl myeloma: Comprasion of effectivity and adverse events
Ali Zahit Bolaman1, Sehmus Ertop2 Atakan Turgutkaya1, Cem Selim, 1 Ayse Hilal Eroglu Kucukerdiler1, Birsen Sahip2, Irfan Yavasoglu1.
1 Adnan Menderes University, School of Medicine, Department of Hematology AYDIN/TURKEY
2 Bulent Ecevit University School of Medicine, Department of Hematology ZONGULDAK/TURKEY
Backround: Lenalidomide is an effective IMID derivative drug in the treatment of patients with multiple myeloma. Lenalidomide is available as original and generic forms in our country. So far, there is no any clinical study comparing generic and original lenalidomine for effectivity and adverse events. We compared generic and original lenalidomide effects and adverse events (AEs) in patients with relapsed multiple myeloma (RMM).
Methods: The patients with RMM using original or generic lenalidomide were evaluated as retrospectively. Overall response (OR), complete response (CR), very good partial response (VGPR), partial response (PR), stable disease and progressive disease rates and also for adverse events, development rates of neutropenia, anemia, thrombocytopenia, febrile neutropenia, anorexia, constipation diarrhea, nausea, vomiting, creatinine increase, transaminase increase, asthenia, fatigue, pyrexia, peripheral edema, upper respiratory system infection, pneumonia, another infection, muscle cramp, back pain, bone pain, muscle weakness, arthralgia, headache, tremor, paresthesia, deep vein thrombosis, pulmonary embolism, hyperglycemia, hypokalemia, hypocalcemia, hypomagnesaemia, skin dry and skin erythema were investigated in myeloma patients. All data were analyzed using the PASW for Windows version 19.0 (SPSS Inc., Chicago, IL, USA). The results were described as a number, frequency, and percentage. The chi-squared test and Fisher's exact test were used for the analysis of categorical data and independence between variables. The results were assessed at 95% confidence interval and p-value of less than 0.05 was accepted as significant.
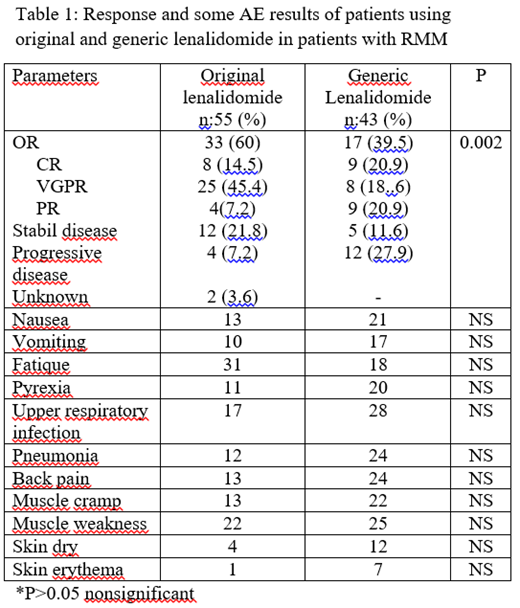
Results: The number of patients using original lenalidomide was 55 and the number of patients using generic lenalidomide was 43. OR rate was 60 % versus 39.5% in patients using original and generic lenalidomide, respectively. CR rate was 14.5%, VGPR was rate 45.4% in original group while CR rate was 20.9 and VGPR 18.6 in patients using generic lenalidomide. AEs were low in original lenalidomide group than generic group. AEs were usually grade 1 or 2. Response and AEs rates are shown in Table 1.
Conclusion: Our study showed original and generic forms of lenalidomide are effective for the treatment of RMM. OR rate was higher in original lenalidomide than generic lenalidomide. The AEs of original lenalidomide were lower than generic lenalidomide without statistically significance. Further studies involving a larger number of patients with RMM would be useful for comparing the efficacy and AEs of original or generic lenalidomide.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal