Background: Novel agent induction and AHCT remains the preferred initial therapeutic strategy for transplant-eligible MM patients. Current prognostic tools in MM focus solely on disease-specific factors at diagnosis to determine patient prognosis-International Staging System (ISS) and revised-ISS (R-ISS). A major limitation to both, the ISS and R-ISS, is that they are not specific for HCT-eligible patients and do not take into account other patient factors that may enter into a decision to pursue AHCT. The data used to generate these staging systems were from broad populations with varying upfront treatment strategies and included patients who were ineligible for intensive therapy. Additionally, there is considerable interest in identifying the population that relapses early despite modern induction/AHCT approaches who are candidates for novel approaches for maintenance/consolidation.
To address these problems, we used data from the Center for Blood and Marrow Transplant Research (CIBMTR) registry to identify disease-, patient-, and transplantation-specific variables that are associated with progression-free survival (PFS) in patients undergoing upfront AHCT (within 12 months of diagnosis).
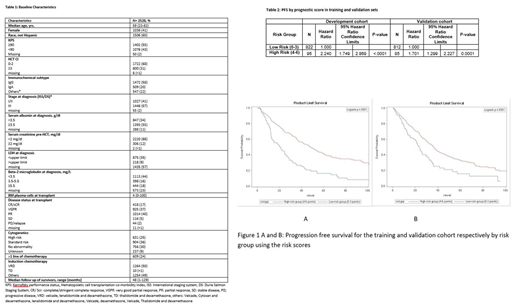
Methods: We used the outcomes of 2528 MM patients undergoing upfront AHCT from 2008-2017 reported to the CIBMTR. Patients were divided into training and validation sets with a 50% random split. High risk cytogenetics was defined as the presence of one or more of the following: t(4;14), t (14;16), t (14;20), del 13q, del 17p, 1q gain, or 1p deletion. We used a Cox multivariable model to identify factors prognostic of progression free survival (PFS) in a training subset. The regression coefficients of the final model was transformed into a risk score with an appropriate transformation. A weighted score using these factors was assigned to the training cohort (n = 917) and validation cohort (n=897) using subset that had all values that entered the final model. Kaplan-Meier estimates of the individual scores were used to classify patients into risk groups for both cohorts.
Results: Baseline characteristics of these patients are shown in Table 1. No cytogenetic abnormality, VRD induction, pre-AHCT bone marrow plasma cells (BMPCs) <10% and 1 line of induction chemotherapy were assigned 0 points. Pre-AHCT BMPCs ≥10% (hazard ratio HR, 1.47; 95% CI, 1.19-1.83), use of ≥2 lines of induction chemotherapy prior to AHCT (HR 1.32; 95% CI 1.06-1.64), standard cytogenetic risk vs. no abnormality (HR 1.41; 95% CI 1.13-1.77) and induction regimens (non-VRD regimens vs. VRD) (HR 1.4, 95% CI 1.17-1.74) were associated with increased hazard of progression and assigned 1 point in the scoring system. Presence of high-risk cytogenetics vs. no abnormality (HR 1.87; 95% CI 1.45-2.42) was assigned 2 points, and the use of thalidomide and dexamethasone (TD) as an induction regimen (HR 2.19; 95% CI 1.48-3.2) was assigned 3 points. A two-category system was created based on the scoring: low risk (0-3) and high risk (4-6). The scoring system was prognostic for PFS when applied to both cohorts. High-risk group was found to have significantly higher risk of progression and/or death compared to low risk in training (HR 2.2; 95% CI 1.74-2.86; p<0.0001) and validation cohort (HR 1.7, 95% CI 1.30-2.22; p=0.0001) respectively (Table 2). The 3-year PFS in the training cohort was 60% (95% CI 56%-64%) in low risk and 27% (95% CI 17%- 36%) in high risk while in the validation cohort was 51% (95% CI 47%-55%) in low risk and 28% (95% CI 16%- 39%) in high risk (Figure 1A and 1B).
Conclusions: We describe a prognostic model specifically for patients undergoing upfront AHCT in MM which can identify patients at very high risk for early relapse/progression. These patients should be ideal candidates for studies of immunotherapy or other interventions after AHCT aimed at reducing relapse.
Dhakal:Sanofi: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; Amgen: Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene: Honoraria; Takeda: Membership on an entity's Board of Directors or advisory committees. Kumar:Celgene: Consultancy, Research Funding; Janssen: Consultancy, Research Funding; Takeda: Research Funding. Shah:Genentech, Seattle Genetics, Oncopeptides, Karoypharm, Surface Oncology, Precision biosciences GSK, Nektar, Amgen, Indapta Therapeutics, Sanofi: Membership on an entity's Board of Directors or advisory committees; Bristol-Myers Squibb: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Nkarta: Consultancy, Membership on an entity's Board of Directors or advisory committees; Kite: Consultancy, Membership on an entity's Board of Directors or advisory committees; Teneobio: Consultancy, Membership on an entity's Board of Directors or advisory committees; University of California, San Francisco: Employment; Poseida: Research Funding; Indapta Therapeutics: Equity Ownership; Celgene, Janssen, Bluebird Bio, Sutro Biopharma: Research Funding. Qazilbash:Amgen: Consultancy, Other: Advisory Board; Autolus: Consultancy; Bioclinical: Consultancy; Genzyme: Other: Speaker. D'Souza:EDO-Mundapharma, Merck, Prothena, Sanofi, TeneoBio: Research Funding; Prothena: Consultancy; Pfizer, Imbrium, Akcea: Membership on an entity's Board of Directors or advisory committees. Hari:AbbVie: Consultancy, Honoraria; Cell Vault: Equity Ownership; Sanofi: Honoraria, Research Funding; Spectrum: Consultancy, Research Funding; Amgen: Research Funding; Kite: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; BMS: Consultancy, Research Funding; Takeda: Consultancy, Honoraria, Research Funding; Celgene: Consultancy, Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal