
Introduction: The direct oral anticoagulants (DOACs: apixaban, dabigatran, edoxaban and rivaroxaban) may be used for the treatment and secondary prevention of venous thromboembolism (VTE) and for the prevention of stroke in the setting of non-valvular atrial fibrillation (NVAF). Adding aspirin (ASA) to a DOAC is often appropriate after acute coronary syndromes or percutaneous coronary intervention. However, many patients receive oral anticoagulation and ASA without a clear need for combination therapy. Current data, largely from patients treated with warfarin and ASA, suggest that the addition of ASA may increase bleeding risk without reducing thrombotic events. However, it is uncertain if this applies to patients on DOACs with ASA, and patients on combination DOAC+ASA therapy were not well represented in clinical trials. We sought to evaluate the impact of adding ASA to DOAC therapy for patients without an apparent indication for combination therapy.
Methods: We conducted a registry-based cohort study of adults on DOAC therapy for NVAF or VTE followed at 6 anticoagulation clinics in Michigan between January 2009 to June 2019, recruited through the Michigan Anticoagulation Quality Improvement Initiative (MAQI2). Patients were excluded if they had a history of heart valve replacement, recent myocardial infarction, or less than 3 months of follow-up. Two propensity matched cohorts (DOACs vs. DOAC+ASA) of patients were analyzed based on aspirin use at the time of study enrollment. The primary outcome was any new bleeding event. Secondary outcomes included new episodes of arterial or VTE, bleeding event type (fatal, life threatening, major, clinically relevant non-major, non-major bleeding (CRNMB), and intracranial or intraspinal), and death. Random chart audits were done to confirm the accuracy of the abstracted data.
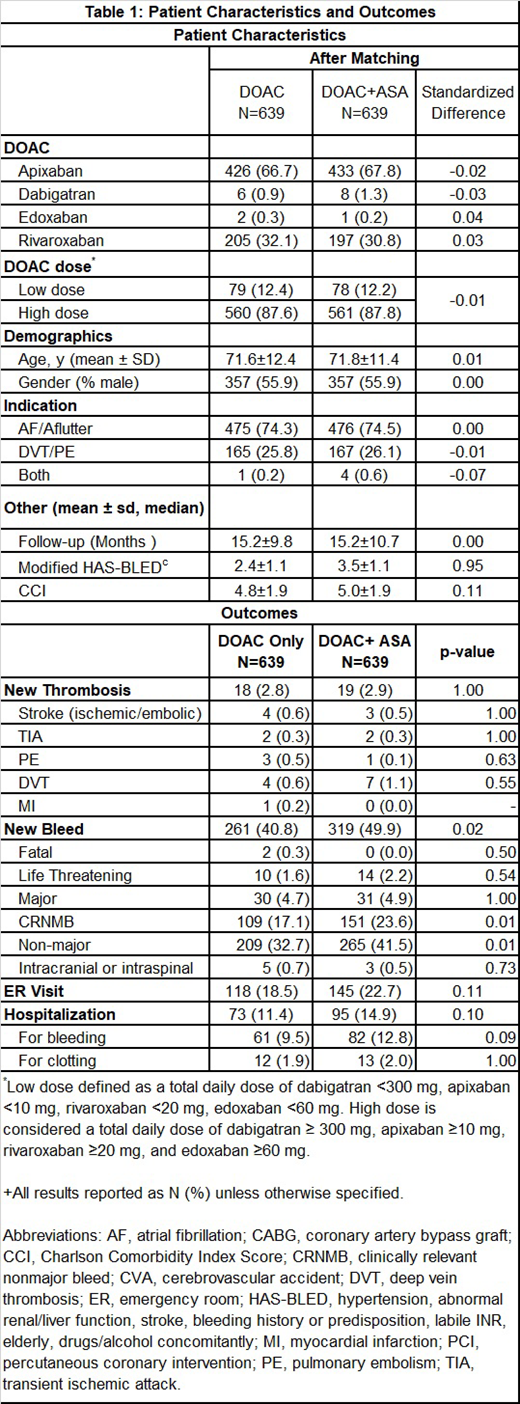
Results:
Of the study cohort of 2,045 patients, 647 (31.6%) patients without a clear indication for ASA received ASA with a DOAC. We compared two groups of 639 matched patients. Patient demographics, co-morbidities, and concurrent medications were well-balanced after propensity score matching. After an average of 15.2 months of follow-up we found that patients on combination therapy (DOAC+ASA) had a significantly higher rate of bleeding compared to patients on DOAC monotherapy (319 bleeding events vs. 261 bleeding events, P=0.02; 0.41 bleeds per patient vs. 0.33 bleeds per patient). This difference was largely driven by CRNMB events (151 with DOAC+ASA vs. 109 with DOAC monotherapy, P=0.01), with the only 2 fatal bleeding events observed with DOAC monotherapy. We did not observe a significant difference between the groups in other bleeding event classifications. Bleeding sites were most commonly cutaneous, gastrointestinal, and genitourinary. Observed rates of thrombosis (stroke, VTE, myocardial infarction, or other) were similar between the groups (19 events with DOAC+ASA vs. 18 events for DOAC monotherapy, P=NS). Patients on combination therapy had more emergency room visits and hospitalizations but these differences were not statistically significant.
Conclusion:
Patients on oral anticoagulation for VTE or NVAF with a DOAC, without a clear need for ASA, experienced more bleeding events with the addition of ASA compared to DOAC monotherapy, without an apparent improvement in the incidence of thrombosis. Further study is needed to assess if DOAC+ASA is safer than warfarin+ASA, to compare the outcomes of the individual DOACs, and to confirm these findings in a larger cohort. Until such assessment is complete, clinicians should carefully consider the need to add aspirin in patients on DOAC therapy.
Kaatz:Portola: Honoraria; Pfizer: Honoraria; Janssen: Honoraria, Research Funding; Bristol Myers Squibb: Honoraria. Kline-Rogers:AC Forum: Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria; QUANTUM-AF: Membership on an entity's Board of Directors or advisory committees. Sood:Bayer: Research Funding. Froehlich:Fibromuscular Dysplasia Society: Research Funding; Janssen: Honoraria; Pfizer: Honoraria; Boehringer-Ingelheim: Honoraria; Merck: Honoraria; Blue Cross Blue Shield of Michigan: Research Funding; Novartis: Honoraria. Barnes:Portola: Honoraria; Janssen: Honoraria; Blue Cross Blue Shield of Michigan: Research Funding; Bristol Myers Squib: Honoraria; AMAG Pharmaceuticals: Honoraria; Pfizer/Bristol Myers Squib: Research Funding; Pfizer: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal