Introduction
Yes-associated protein (YAP) is an important transcriptional regulator and effector of the Hippo signaling that has emerged as a novel determinant of malignancy in several human tumors. Verteporfin (VP), a clinically used photosensitizer for treatment of macular degeneration, is recently identified as a potential inhibitor of YAP expression independent of light activation. However, the function and mechanism of Hippo-YAP in diffuse large B-cell lymphoma (DLBCL) remains ill defined. Herein, we investigated the functional significance of YAP and proposed a novel therapeutic strategy for DLBCL.
Methods
Lymph node biopsies from 60 de novo DLBCL patients and 30 reactive hyperplasia cases were collected with informed consents. The biological function of YAP was evaluated via RNAi-mediated knockdown and CRISPR/Cas9 mediated genomic deletion. RNA-sequencing was conducted to detect the dysregulated RNAs in DLBCL cell with YAP knockout. Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways analyses were used to explore the function of these differentially expressed RNAs. SCID-Beige mice were subcutaneously injected with DLBCL cells to establish xenograft model. Animal experiments were performed in accordance with the principles of the Institutional Animal Care.
Results
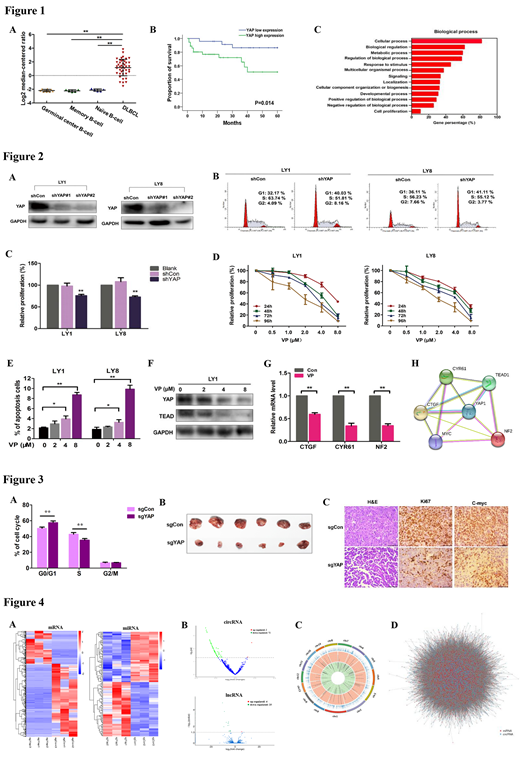
We first examined the expression of YAP in Oncomine database and discovered the upregulation of YAP mRNA in DLBCL cells (Fig.1A). High expression of YAP protein was validated in a cohort of newly diagnosed DLBCL patients (n=60). Survival analysis revealed that YAP expression was associated with aggressive disease process (p=0.014, Fig.1B).
Functional enrichment analyses of YAP in DLBCL microarray profiles revealed that YAP was enriched in cellular process and biological regulation (Fig. 1C). Knockdown of YAP (shYAP) significantly suppressed cell proliferation and promoted cell cycle arrest (Fig.2A-C). VP restrained proliferation of DLBCL cells in a dose- and time-dependent manner (Fig. 2D). Treatment of VP significantly promoted cell apoptosis and cell cycle arrest in DLBCL cells (Fig. 2E). We next validated the involved mechanism of the anti-tumor effect of VP in DLBCL cells. Protein expression levels of YAP and TEAD were significantly inhibited by VP in dose-dependent manner (Fig.2F). In addition, treatment of VP remarkably restrained the mRNA expression of YAP targeted genes, including CTGF, CYR61 and NF2 (Fig. 2G). Genomic network enrichment of YAP interactions was established in STRING database (Fig. 2H).
To validate the involvement of YAP in DLBCL pathogenesis, we deleted YAP expression by CRISPR/Cas9 genomic-editing system. YAP deletion (sgYAP) resulted in significantly reduction in cell proliferation and arrest of cell cycle (Fig.3A). To explore the activities of YAP inhibition in vivo, xenograft DLBCL mice model was established by LY1 cells with YAP-deletion. Compared to the control group, tumors with YAP-deletion displayed reduced growth and decreased expression levels of Ki67 and C-myc (Fig. 3B-C).
To further explore the translational regulation mechanism of YAP in DLBCL, integrated analysis of mRNA and non-coding RNA (ncRNA) expression profiles were conducted by whole-transcriptome sequencing (RNA-seq). 158 miRNAs, 29 lncRNAs, 74 circRNAs and 528 mRNAs were shown to be significantly differentially expressed (DE) in DLBCL cells with YAP knockout (Fig. 4A-C). The subsequent GO and KEGG pathway analysis revealed that DE molecules mainly related to cellular metabolic process and pathways. In addition, the miRNA-circRNA network was established (Fig. 4D).
Conclusion
Taken together, these findings provide in vitro and in vivo pre-clinical evidence for the crucial role of YAP in lymphomagenesis and highlight that blockade of YAP represents an attractive approach in DLBCL. The clinical used photosensitizer Verteporfin exerted anti-tumor effect via disrupting YAP-TEAD complex. Further interrogation on the regulatory mechanism of YAP in DLBCL will outline a promising therapeutic option to utilize this newly identified oncogene in DLBCL therapy.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal