Introduction: Immunomodulatory drugs (IMiDs) are being frequently used as an integral part of induction regimen as well as maintenance after Bone Marrow Stem cell Transplant (BMSCT) therapy as backbone of therapy for patients with plasma cell neoplasm (PCN) including Multiple Myeloma (MM). Therapy is often complicated by dermatologic adverse effects which may herald a favorable prognosis suggesting a more robust immune stimulation by this class of drugs. The incidence of IMiD-associated rash is up to 27% in some reports and can range from mild to moderate to severe and life threatening skin toxicity including Steven-Johnsons syndrome and toxic epidermal necrolysis. IMiD-associated skin eruptions are thought to result from differential T cell immune modulation. The optimal management strategy for IMiD induced rash is unknown. The European Myeloma Network recommends topical corticosteroids and antihistamines for localized skin reactions. For severe reactions, a desensitization protocol with prolonged 6-week steroid taper is recommended based on a case series of five patients that developed delayed cutaneous adverse effects to IMiD [Lee MJ et al]. Dexamethasone used concomitantly with IMiD as part of the treatment regimen does not decrease the occurrence of skin rash as shown by Sviggum et al. The likely explanation is that concurrent dexamethasone is not effectively able to exert immunosuppressive activity to mitigate the occurrence of cutaneous reactions. Hence, a prolonged, daily steroid protocol is required for effective desensitization. Therefore, we designed a standardized 3-week steroid rash prophylaxis protocol with a low dose daily corticosteroid tapering regimen that allows desensitization and reinstitution of the same IMiD. We assessed the impact of this desensitization regimen on clinical outcomes, by comparing patients with versus without dermatologic manifestations.
Methods: Dermatologic adverse effects associated with IMiDs in our study were managed by a single oncologist using a standardized approach. A total of 87 patients were evaluated, 24 patients in the rash group vs 63 controls. A cohort of age- and gender-matched without rash (n = 63) was randomly selected from the institutional database. The effects of rash on overall and progression free survival (OS and PFS) were further estimated using Cox regression controlling for the effects of age and gender.
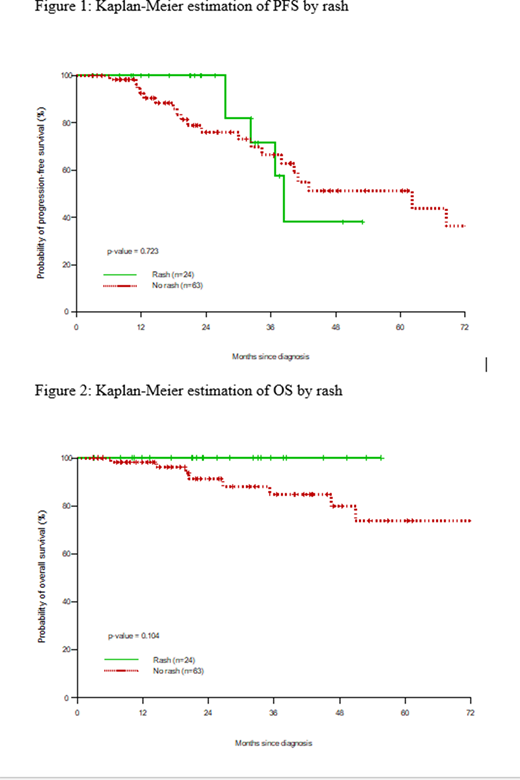
Results: Median time to development of rash after IMiD initiation was 28 days (range 2-232 days). The incidence of rash was 27.6% (95% CI: 19.3%-37.8%). All patients were managed by temporary treatment interruption and upon clearance of rash, re-institution of the same IMiD concomitantly with a standardized 3-week steroid rash prophylaxis protocol (prednisone at 10 mg daily for 10 days, followed by 5 mg daily for 10 days, followed by 5 mg on alternate days for 10 days). As a result, all patients were able to restart the same IMiD with none re-experiencing any dermatologic adverse effect afterward. Comparing to controls without rash, there was no significant difference in PFS (p=0.769) or OS (p=0.24) in the multivariable regression model.
Conclusions: Proposed 3-week corticosteroid regimen showed 100% success rate in re-instituting IMiDs without recurrence of skin rashes in our cohort. The results of our study indicate that the 30-day prednisone course is practical, well tolerated, effectively desensitizes and allows re-institution of IMiD enabling patients to enjoy comparable outcomes to those without skin rash. Whether development of rash correlates with improved outcomes is unknown. Here, outcomes were similar in both cohorts.
Caimi:Celgene Corp: Other: Incyte Corporation - Ownership - Pharmacyclics, Inc. - Ownership - Celgene Corp. - Other, Speakers Bureau; ADC Therapeutics: Research Funding; Genentech: Research Funding. de Lima:BMS: Other: Personal Fees, advisory board; Celgene: Research Funding; Pfizer: Other: Personal fees, advisory board, Research Funding; Incyte: Other: Personal Fees, advisory board; Kadmon: Other: Personal Fees, Advisory board. Malek:Bluespark: Research Funding; Takeda: Other: Advisory board , Speakers Bureau; Medpacto: Research Funding; Cumberland: Research Funding; Amgen: Honoraria; Clegene: Other: Advisory board , Speakers Bureau; Sanofi: Other: Advisory board; Janssen: Other: Advisory board, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal