Background:
Myelofibrosis (MF) is a Philadelphia chromosome negative myeloproliferative neoplasm associated with systemic and splenomegaly-related symptoms, cytopenias and decreased survival. Approval of ruxolitinib, an oral janus kinase (JAK)-inhibitor, for higher-risk MF patients (pts) by the Food and Drug Administration in 11/ 2011 opened a new era of targeted treatment for this disease. There are limited data on the "real-world" clinical experiences and outcomes of pts with MF treated in the JAK inhibitor era. MF became reportable to population-based cancer registries including the Surveillance, Epidemiology and End Results (SEER) program in 2001, making its investigation possible at the population level. The objective of this study was to assess the patterns of care and outcomes of older MF pts in the ruxolitinib era.
Methods:
Using the linked SEER-Medicare database, we identified a cohort of older pts diagnosed with MF from 2007 through 2015 who fulfilled the following eligibility criteria: 1) aged 66-99 years at diagnosis; 2) had known month of diagnosis; 3) were not identified from death certificate or autopsy only; 4) had continuous enrollment in Medicare Parts A, B and no enrollment in health maintenance organizations from 1 year before diagnosis until the end of follow-up (death or 12/31/2016, whichever came first); 5) had continuous enrollment in Medicare Part D from diagnosis until the end of follow-up; and 6) bone marrow biopsy claim from 1 year before diagnosis to end of follow up.
Treatments were assessed via Medicare parts B&D claims. Kaplan-Meier curves and log-rank tests were used to compare survival between patient groups. Multivariable cox proportional hazards regression models were used to assess the effect of ruxolitinib use on survival in MF pts. Aside from treatment, we considered the influence of several characteristics on survival, including age at diagnosis, sex, race/ethnicity, marital status, comorbidities, SEER region and percentage living in poverty at the census tract level.
Results:
Among 528 MF pts, median age at diagnosis was 76 (interquartile range [IQR], 71- 80) years with 88.8% white and 56.1% male. 230 pts were diagnosed in the early era (2007-2011), and 298 in the late era (2012-2015), of which 113 (37.9%) were ruxolitinib users. There was no difference among any evaluated characteristics between two eras and by ruxolitinib status in the late era.
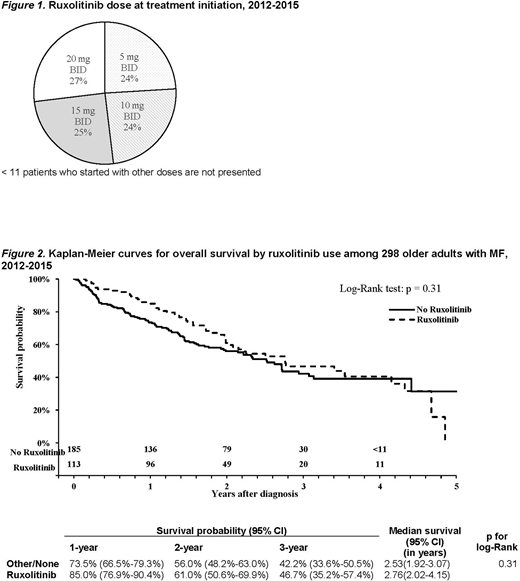
The median duration of ruxolitinib use was 11.9 months. Similar number of pts started at 5, 10, 15 and 20 mg twice a day (BID) (Figure 1). Among 31 pts who started at ≤5 mg BID, 15 (48.4%) never had their dose of ruxolitinib escalated. While on ruxolitinib treatment, nearly half of the pts received additional medications for symptom management including hydroxyurea (22.6%), prednisone (17.9%) or both (10.4%). < 11 users were able to go up to the highest dose of 25 mg BID. Ruxolitinib was interrupted > 30 days for 31 times by 20 of 113 (17.7%) pts with median interruption duration of 43 (IQR 34-71) days.
The median survival was 2.70 (95% confidence interval [CI]: 1.87-3.41) years and 2.62 (95% CI: 2.15-3.07) for the early and late era pts, respectively (p for log-rank 0.91). The multivariable analysis showed no impact of diagnosis era on survival (late vs early era hazard ratio (HR) of 1.08, 95% CI 0.83-1.40; p= 0.57). There was no difference in survival by ruxolitinib status (log-rank test, p=0.31), with a median survival of 2.76 (95% CI: 2.01-4.15) years and 2.53 (95% CI: 1.92-3.07) years among users and non-users, respectively (Figure 3). In the multivariable analysis, the risk of death among ruxolitinib users compared to non-users was not statistically significant with HR of 0.82 (95% CI 0. 59-1.16; p= 0.26).
Conclusions:
Older MF pts treated with ruxolitinib had similar survival when compared to pts who did not receive this medication, but the choice of ruxolitinib might have been influenced by disease risk which we were unable to assess. For many ruxolitinib users, the drug was interrupted, the dose was not escalated, additional medications were used concurrently (possibly to help control disease manifestation), and treatment was discontinued quickly after initiation. Optimization of ruxolitinib use may be necessary to accomplish better outcomes. Furthermore, development of new drugs which may be used together with ruxolitinib or after its discontinuation is needed.
The work was supported by The Frederick A. Deluca Foundation.
Wang:Celgene/BMS: Research Funding. Ma:Celgene/BMS: Research Funding; BMS: Consultancy. Podoltsev:Jazz Pharmaceuticals: Research Funding; Celgene: Consultancy, Honoraria, Research Funding; Agios Pharmaceuticals: Consultancy, Honoraria; Sunesis Pharmaceuticals: Research Funding; Pfizer: Consultancy, Honoraria, Research Funding; Blueprint Medicines: Consultancy, Honoraria; Bristol-Myers Squib: Consultancy, Honoraria; Genentech: Research Funding; AI Therapeutics: Research Funding; Samus Therapeutics: Research Funding; Astellas Pharma: Research Funding; Kartos Therapeutics: Research Funding; CTI biopharma: Consultancy, Honoraria, Research Funding; Boehringer Ingelheim: Research Funding; Novartis: Consultancy, Honoraria; Alexion: Consultancy, Honoraria; Incyte: Consultancy, Honoraria; Astex Pharmaceuticals: Research Funding; Daiichi Sankyo: Research Funding; Arog Pharmaceuticals: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal