Immunoglobulin light Chain (AL) amyloidosis is the most common form of systemic amyloidosis, accounting for approximately 70% of the diagnosed cases in developed countries. There are no approved treatments for AL amyloidosis, and currently autologous stem cell transplants and/or chemotherapy targeted to eradicate the underlying plasma-cell dyscrasia (PCD) to stop the production of misfolded light chains are used. No therapies are available to remove existing amyloid deposits, improve and/or reverse organ dysfunction.
CAEL-101 is a chimeric immunoglobulin G1 kappa isotype that reacts specifically with light chain fibrils, irrespective of their κ or λ isotype but, notably, not with the native forms. CAEL-101 was shown to bind to a cryptic epitope at the N-terminal of light chain proteins that adopt a non-native b-sheet structure, which is conserved in κ and λ mis-folded light chains. It is hypothesized that CAEL-101 will modify the disease course of AL amyloidosis by facilitating the removal of amyloid fibrils deposited in tissues. CAEL-101 was previously studied as monotherapy in an open label dose escalation Phase 1 study where it was shown to be safe, tolerable up to 500 mg/m2 and associated with an early organ response. No DLT were seen and the PK profile appeared not to reach linearity.
The current multicenter, open-label, sequential cohort, dose-selection study of CAEL-101 in Mayo Stage I, II and IIIa AL amyloidosis patients was to define the safety and tolerability of CAEL-101 in combination with Cyclophosphamide-Bortezomib-Dexamethasone (CyBorD) as the standard of care (SOC) PCD therapy during a 27-day treatment period and to determine the recommended dose for the subsequent Phase 3 studies. Patients had measurable hematologic disease, as defined by at least one of:
involved/uninvolved free light chain (FLC) difference (dFLC) > 5mg/dL or
FLC >5mg/dL with abnormal ratio or
serum protein electrophoresis (SPEP) m- spike > 0.5 g/dL
Patients were seen in the clinic every week for 4 weeks to receive CAEL-101 2-HR IV infusions, and for safety and tolerability assessments. Subsequently, patients have been receiving CAEL-101 infusions approximately every other week.
Actual dose was determined by patient body surface area in meters squared. When administered on the same day, CAEL-101 was administered first before CyBorD chemotherapy.
The study employed a 3+3 dose escalation design, starting with a 500 mg/m2 dose. Patients were followed for dose-limiting toxicities (DLTs). The DLT observation period for the first cohort was through 14 days following the first infusion. For subsequent cohorts, this was through 27 days. At least 3 patients were enrolled in each dose cohort. Enrollment into a new cohort with the next higher dose of CAEL-101 did not begin until the DLT observation period had been completed for the last patient enrolled in the previous cohort, with further dosing cohorts at 750 and 1000 mg/m2. Safety assessment included vital signs, physical examinations, electrocardiograms (ECGs), clinical laboratory parameters (hematology, serum chemistry, urinalysis), and TEAEs. The pharmacokinetic (PK) profile of CAEL-101 was also assessed.
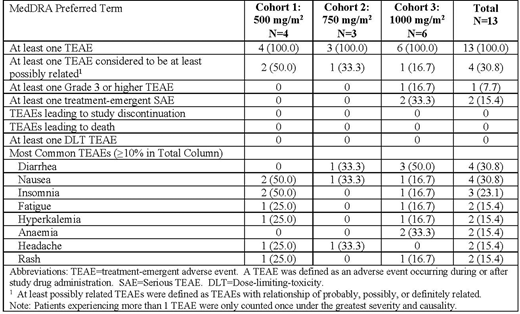
The 13 patients averaging 65.2 years (range 47.6 to 79.6 years). The majority were male (76.9%), white (84.6%), and non-Hispanic (100%). Mayo staging I (7.7%), II (69.2%), and III (23.1%) reflected a wide range of disease severity in the patients enrolled. All patients were treated successfully through their 4th dose, the highest (1000 mg/m2) cohort enrolling 6 patients. No DLT was observed, and CAEL-101 was well-tolerated by all patients. The most common TEAEs were diarrhea and nausea (each 30.8%). Dose-normalized PK concentrations were best described by a linear two-compartment model, with terminal half-life of 28 days.
This study demonstrated that CAEL-101 at doses up to 1000 mg/m2, given with CyBorD, was well-tolerated in the AL amyloidosis population. Two Phase 3 efficacy/safety studies, one enrolling Mayo Stage IIIa and the other Stage IIIb patients, have been initiated, and based on the findings from this Phase 2 study, the 1000 mg/m2 dose of CAEL-101 is being used for treatment in those studies.
Valent:Takeda Pharmaceuticals: Other: Teaching, Speakers Bureau; Celgene: Other: Teaching, Speakers Bureau; Amgen Inc.: Other: Teaching, Speakers Bureau. Silowsky:Caelum Biosciences: Current Employment, Current equity holder in private company. Kurman:Caelum Biosciences: Consultancy. Daniel:Caelum Biosciences: Current Employment, Current equity holder in private company. Jobes:Caelum Biosciences: Current Employment, Current equity holder in private company. Harnett:Caelum Biosciences: Consultancy, Current equity holder in private company. Ziola:Caelum Biosciences: Consultancy. Roviwong:Caelum Biosciences: Current Employment, Current equity holder in private company. Spector:Caelum Biosciences: Current Employment, Current equity holder in private company. Sobolov:Caelum Biosciences: Current Employment, Current equity holder in private company.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal