Introduction
The pathogenesis of classic Hodgkin's lymphoma (cHL) is largely unsettled. Previous studies have provided limited support to the possible association between tobacco smoking and outcome in cHL with inadequate evidence of a dose-outcome relationship. Smoking can result in a multitude of preventable malignancies, thus we studied the association between smoking, the initial stage of cHL and survival outcomes.
Methods
This single-center study included patients with a diagnosis of cHL who were first seen at The University of Texas MD Anderson Cancer Center between January 1,2016 and May 28, 2020 for either newly diagnosed cHL or relapsed/refractory (R/R) cHL.
Patients' charts were reviewed to assess the history of active smoking, former smoking and the amount of smoking (measured as pack-year). Former smoking was defined as history of smoking prior to the diagnosis of cHL and no active smoking at the time of diagnosis. The primary aims were to assess overall survival (OS), progression-free survival (PFS) and frequency of advanced disease at the time of diagnosis. Descriptive statistics including mean, standard deviation, median, and range for continuous variables such as age, amount of smoking and lab measurements, and frequency counts and percentages for categorical variables such as race, gender, history of current/prior smoking and response were analyzed. Kaplan-Meier method was used for time-to-event analysis including PFS and OS. Median time to event in months with 95% confidence interval (CI) was calculated. The Log-rank test was used to evaluate the difference in time-to-event endpoints between patient groups. Statistical software SAS 9.4 (SAS, Cary, NC) and S-Plus 8.2 (TIBCO Software Inc., Palo Alto, CA) were used for statistical analyses.
Results
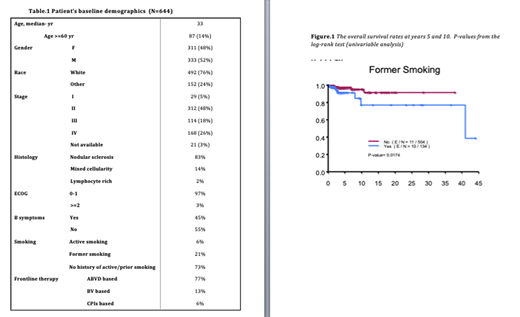
Between 2016 and 2020, 644 patients met the inclusion criteria. The median age at diagnosis was 33 years (range: 9-85 years) with 52% males. Advanced stage (stages III and IV) occurred in 45% of patients. Ninety-seven percent of patients had 0-1 Eastern Cooperative Oncology Group (ECOG) performance status. Histological subtype was predominately nodular sclerosis (83%). International Prognostic Index was =>4 in 13% of patients. Most of the patients received doxorubicin, bleomycin, vinblastine and dacarbazine (ABVD) based therapy (77%), brentuximab vedotin (BV) based therapy (13%)and checkpoint inhibitors (CPIs) based therapy (6%). Patient's demographics are outlined in Table.1. The percentage of active smoking was low at 6% (median age of active smokers: 33.5). History of prior smoking occurred in 21% of patients. The mean amount of smoking was 1.87 pack-year (rang :0. 05-54 pack-year). Active smoking was associated with advanced stage (9.5% vs. 4.9%, p-value: 0.033). PFS rate at 5 years was similar in smokers and non smokers (52% vs. 54%, p-value 0.9). OS rate at 10 years was better in never smokers when compared to former smokers (95% vs. 77%, p-value: 0.017). Univariable Cox proportional hazards model for OS showed a significant association between amount of smoking and OS with hazard ratio of 1.04 (95% CI: 1.005 1.07, p-value: 0.025).
Conclusions
We report the largest analysis of smoking status and impact on advanced stage and cHL clinical outcomes. Smoking history is associated with inferior 10 year OS (18% lower OS in patients with history of former smoking). Active smoking was associated with advanced stage however the frequency of active smoking in our patient database was low at 6% compared to previous reports of higher incidence of up to 20%. Patients should be counseled against smoking to avoid worse outcome associated with smoking in many clinical conditions including cHL.
Parmar:Cellenkos Inc.:Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees, Patents & Royalties, Research Funding.Iyer:Legend Biotech:Consultancy;CRISPR:Research Funding;Rhizen:Research Funding;Curio Biosciences:Honoraria;Target Oncology:Honoraria;Afffimed:Research Funding;Merck:Research Funding;Spectrum:Research Funding;Trillium:Research Funding;Daiichi Sankyo:Consultancy;Seattle Genetics, Inc.:Research Funding.Nieto:Astra Zeneca:Other: Grant Support;Secura Bio:Other: Grant Support;Affimed:Consultancy, Other: Grant Support;Novartis:Other: Grant Support.Chuang:Sage-Evidence=Based Medicine & Practice:Consultancy.Wang:Guidepoint Global:Consultancy;Dava Oncology:Honoraria;Loxo Oncology:Consultancy, Research Funding;Verastem:Research Funding;Beijing Medical Award Foundation:Honoraria;Targeted Oncology:Honoraria;OMI:Honoraria, Other: Travel, accommodation, expenses;AstraZeneca:Consultancy, Honoraria, Other: Travel, accommodation, expenses, Research Funding;OncLive:Honoraria;Molecular Templates:Research Funding;Celgene:Consultancy, Other: Travel, accommodation, expenses, Research Funding;Kite Pharma:Consultancy, Other: Travel, accommodation, expenses, Research Funding;Pulse Biosciences:Consultancy;MoreHealth:Consultancy;Janssen:Consultancy, Honoraria, Other: Travel, accommodation, expenses, Research Funding;Pharmacyclics:Consultancy, Honoraria, Other: Travel, accommodation, expenses, Research Funding;BioInvent:Research Funding;Lu Daopei Medical Group:Honoraria;Juno:Consultancy, Research Funding;VelosBio:Research Funding;Acerta Pharma:Research Funding;InnoCare:Consultancy;Oncternal:Consultancy, Research Funding;Nobel Insights:Consultancy.Lee:Celgene:Research Funding;Seattle Genetics:Research Funding;Bristol-Myers Squibb:Consultancy, Research Funding;Aptitude Health:Speakers Bureau;Guidepoint Blogal:Consultancy;Takeda:Research Funding;Oncternal Therapeutics:Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal