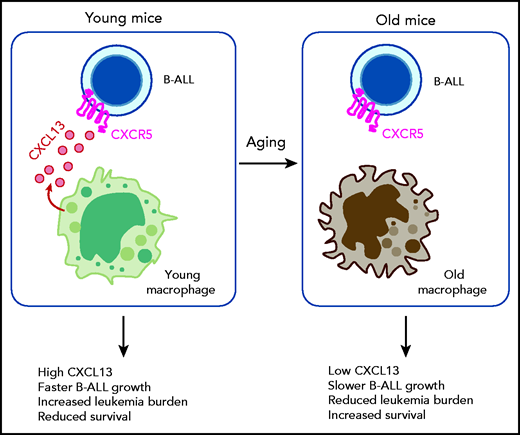
In this issue of Blood, Zanetti et al1 use mouse models of B-cell acute lymphoblastic leukemia (B-ALL) to demonstrate that CXCL13 produced by macrophages in the bone marrow microenvironment activates CXCR5 in B-ALL leukemia cells to drive disease progression. Notably, this pathway was present in young mice but was absent in old mice, indicating that the age of the microenvironment profoundly influences the progression of B-ALL.
The microenvironment is the collection of nonhematopoietic (eg, perivascular stromal cells and endothelial cells) and hematopoietic cells that supports and regulates blood cell production in the bone marrow.2 Macrophages are key components of the bone marrow microenvironment, and several subtypes of macrophages have been identified with unique functions in regulating stromal cells, erythrocyte maturation, and even hematopoietic stem cells directly.2,3 The bone marrow microenvironment (including macrophages) is profoundly perturbed, leading to loss of supportive cells and cytokines, and subverted, to promote disease, by leukemia cells. Disrupting the interactions between the microenvironment and leukemic cells can protect from and delay disease in murine models of leukemia.4,5
For reasons that are not well understood, most cases of B-ALL occur in children.6 Changes in the microenvironment caused by aging promote the expansion of myeloid cells and myeloid-biased stem cells.7 Zanetti et al hypothesized that the bone marrow microenvironment in the young may be more supportive of B-ALL and may explain the high incidence of B-ALL in children. To test this hypothesis, they transplanted B-ALL cells, generated by retroviral transduction of the BCR-ABL1 oncogene in vitro, into young or old mice. They found a two-fold increase in leukemia burden and reduced survival in the young cohort. Notably, they found no differences in tumor burden between young and old mice that received transplants in a model of chronic myeloid leukemia that is also driven by the BCR-ABL1 oncogene. These experiments formally demonstrated that young mice are more susceptible to B-ALL and that this susceptibility is dependent on non–cell-autonomous pathways. Using in vivo confocal immunofluorescence of the bone marrow, Zanetti et al showed that B-ALL cells transplanted into young mice were more motile and localized closer to the endosteum when compared with the same B-ALL cells transplanted into old mice. This finding strongly suggests that the local bone marrow microenvironment differentially regulates B-ALL behavior in young and old mice.
B-ALL primarily affects pediatric patients. Zanetti et al demonstrate that CXCL13 produced by bone marrow macrophages in young mice activates CXCR5 in B-ALL cells, leading to fast leukemia growth and poor survival. Physiological aging results in the loss of the CXCL13-CXCR5 axis, leading to slower leukemia growth and improved survival. Their study demonstrates that the age of the microenvironment controls leukemia outcomes.
B-ALL primarily affects pediatric patients. Zanetti et al demonstrate that CXCL13 produced by bone marrow macrophages in young mice activates CXCR5 in B-ALL cells, leading to fast leukemia growth and poor survival. Physiological aging results in the loss of the CXCL13-CXCR5 axis, leading to slower leukemia growth and improved survival. Their study demonstrates that the age of the microenvironment controls leukemia outcomes.
Since macrophages are known to support leukemia growth,5 Zanetti et al purified and cultured F4/80+CD169+CX3CR1− macrophages from the marrow of young and old mice. In coculture experiments, young macrophages attracted B-ALL cells and promoted their proliferation when compared with macrophages purified from old mice. This effect was mediated by a soluble molecule, given that conditioned media from young, but not old, macrophages recapitulated the phenotype. Old macrophages displayed reduced proliferation and increases in reactive oxygen species, DNA double-strand breaks, and mitochondrial stress when compared with macrophages purified from young mice. These factors correlated with changes in gene expression and chromatin accessibility and suggest that aging results in an overall reduction of macrophage fitness and inflammatory potential.
Because young macrophages promoted B-ALL cell proliferation via a soluble factor and CXCL13 is a known B-cell chemoattractant, Zanetti et al focused on this chemokine. They found that young macrophages produced more CXCL13 than old macrophages and that CXCR5, the receptor for CXCL13, was more highly expressed in B-ALL cells transplanted into young recipients. Inhibition or blockade of CXCL13 reduced growth of B-ALL cocultured with young, but not old, macrophages, whereas CXCL13 treatment was sufficient to promote faster B-ALL growth in vitro and in vivo. In agreement, mouse recipients of Cxcr5−/− B-ALL cell transplants had reduced leukemic burden and increased survival when compared with those receiving wild-type leukemia cells. Furthermore, systemic depletion of macrophages and other phagocytes with clodronate-loaded liposomes reduced the leukemic burden and prolonged survival exclusively in young mice. These rigorous experiments demonstrate a key role for the CXCL13-CXCR5 axis in B-ALL and that this pathway is only functional in the young bone marrow microenvironment, most likely via CXCL13-producing macrophages (see figure). Therefore, the age of the microenvironment controls B-ALL progression.
The aging of the hematopoietic system results in extensive remodeling of the microenvironment, with changes in the frequencies and function of its cellular components and redistribution of hematopoietic stem cells within.7,8 Zanetti et al demonstrate that this remodeling directly leads to changes in the biology of the leukemia. We anticipate that aging-induced remodeling of the microenvironment also affects leukemia initiation, progression, and chemotherapy resistance in other hematological malignancies and provides a rationale for some of the differences observed between leukemias in pediatric and geriatric patients.
Is this CXCL13-CXCR5 proleukemic pathway also functional and significant in humans? Zanetti et al found increased frequency of CD68+ macrophages and CXCL13 protein in bone sections from pediatric patients when compared with adult patients with B-ALL. In retrospective analyses of published B-ALL patient cohorts, they found that low CXCR5 levels correlated with better outcomes for some B-ALL subsets. The major limitation of these analyses is that the number of patients examined is too small to draw firm conclusions. Nevertheless, these findings provide a strong rationale for investigating whether CXCR5/CXCL13 expression can be used as predictive markers and whether this axis can be therapeutically targeted in B-ALL.
In addition to its direct clinical implications, the findings by Zanetti et al also raise exciting new questions. What is the role of CXCL13-producing macrophages in the young bone marrow? How is CXCL13 production, or CXCL13-producing macrophages, lost during aging? Answering these is a necessity, to ascertain whether this macrophage pathway can be therapeutically blocked without deleterious effects on hematopoiesis.
Conflict-of-interest disclosure: The authors declare no competing financial interests.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal