Abstract
Background: In January 2021, the Dutch vaccination programme against SARS-CoV-2 was started. Clinical studies have shown that systemic reactions occur in up to 50% of vaccine recipients. Therefore, COVID-19 vaccination could affect anticoagulation control, potentially leading to an increased risk of thrombotic events and bleeding complications.
Aims: To investigate whether the BNT162b2 vaccine affects anticoagulation control in outpatients using Vitamin K antagonists (VKAs).
Methods: A case-crossover study was performed in a cohort of outpatient VKA users from four Dutch anticoagulation clinics who received a BNT162b2 vaccine. INR results and VKA dosages before the first vaccination, the reference period, were compared with those after the first and second vaccination.
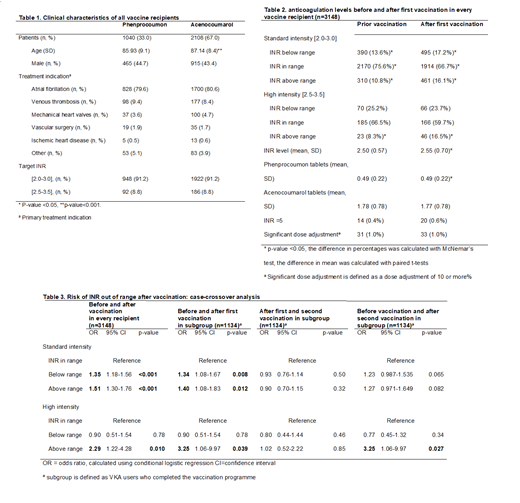
Results: A total of 3148 outpatient VKA users were included, with a mean age (standard deviation (SD)) of 86.7 (8.7) years, of whom 43.8% were male, 67.0% used acenocoumarol and 33.0% phenprocoumon. We observed a decrease of 8.9% of INRs within range in the standard intensity group (target INR 2.0-3.0). There was both an increased risk of supratherapeutic [OR=1.34 (95% CI 1.08-1.67)] and subtherapeutic levels [OR=1.40 (95% CI 1.08-1.83)] after first vaccination. In the high-intensity group (target INR 2.5-3.5), the risk of a supratherapeutic INR was 2.3 times higher after first vaccination [OR=2.29 (95% CI 1.22-4.28)] and 3.3 times higher after second vaccination [OR 3.25 (95% CI 1.06-9.97).
Conclusion: BNT162b2 was associated with an immediate negative effect on anticoagulation control in patients treated with vitamin K antagonists, so it is advisable to monitor the INR short after vaccination, even in stable patients.
Kruip: Daiichi Sankyo: Research Funding; Bayer: Honoraria, Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal