Abstract
Background:
Bispecific anti-CD20, anti-CD19 (LV20.19) CAR T-cells may improve outcomes in relapsed, refractory (R/R) non-Hodgkin lymphoma (NHL) by limiting relapse from single antigen downregulation. We previously reported outcomes of a phase I trial of LV20.19 CAR T-cells expanded in IL-2 in R/R NHL & CLL with high response rates (Shah Nat Med 2020). Preclinical models have shown that, compared to IL-2, CAR T-cells expanded in IL-7 & IL-15 (IL7+15) have improved expansion, in-vivo persistence, and anti-tumor efficacy (Xu Blood 2014). To optimize our CAR-T product, we opened a phase I/II trial of LV20.19 CAR T-cells expanded in IL-7+15 and manufactured under variable lengths of time (8 vs 12 days) in R/R NHL (NCT04186520). We hypothesized that expansion in IL7+15 and manufacturing for shorter duration would generate a CAR-T product with superior functionality.
We contrast comprehensive functional profiling of LV20.19 CAR T-cells expanded in IL7+15 vs IL-2 and manufactured for 8 vs 12 days using single cell cytokine analysis (Isoplexis single cell proteomics system).
Methods:
LV20.19 CAR T-cells were manufactured using the CliniMACS Prodigy device (Shah Nat Med 2020). CAR T-cells were thawed, CD4 & CD8 cells sorted via immunomagnetic separation, and stimulated with CD19+ K562 cells. Stimulated cells were loaded onto single-cell Adaptive Immune Isocode chips and read in an Isolight instrument. The single cell production of 32 cytokines were measured. Polyfunctionality (PFA) and a polyfunctional strength index (PSI) were calculated for each CAR-T product. PFA was defined as a T cell that produced ≥2 cytokines upon stimulation with CD19+ cells. PSI was defined as the % of polyfunctional T cells relative to all T cells, multiplied by the sum of the mean fluorescence index of each assayed cytokine from the polyfunctional T cells. Variables were analyzed with parametric or non-parametric tests where appropriate. A P value <.05 was considered significant and all P values are 2-tailed.
Results:
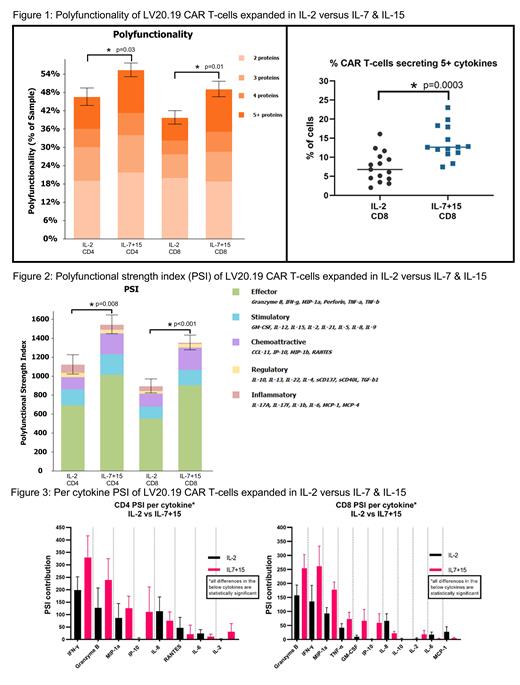
There were 14 patients from the IL7+15 trial and 15 from the IL-2 trial included in the analysis. For cells expanded in IL7+15 vs IL-2, global PFA (52.3% vs 43.2%, p=0.001), CD4 PFA (55.4% vs 46.6%, p=0.03) and CD8 PFA (49.1% vs 39.8%, p=0.01) were all significantly higher as was the proportion of cells secreting ≥5 cytokines (figure 1). PSI was significantly higher for IL7+15 vs IL-2 expanded cells, both globally (1452 vs 1011, p<0.001) and for CD4 (1548 vs 1125, p=0.008) and CD8 T-cell subsets (1356 vs 897, p<0.001) (figure 2). In assessing contribution to the PSI per individual cytokine, IL7+15 vs IL-2 expanded CD4 cells had significantly higher levels of IFN-γ, granzyme B, MIP-1a, IP-10, and IL-2 and less IL-8, RANTES, and IL-6 (figure 3). IL7+15 vs IL-2 expanded CD8 cells had significantly higher levels of granzyme B, IFN-γ, MIP-1a, TNF-α, GM-CSF, IP-10 and IL-2, and less IL-8, IL-10, IL-6 and MCP-1.
In the IL7+15 trial, there were 8 patients included in the 8-day manufacturing arm and 6 patients in the 12-day arm. For the 8- vs 12-day arms, global PFA (54.1% vs 54.2%, p=0.98), CD4 PFA (56.3% vs 54.2%, p=0.70) and CD8 PFA (51.8% vs 45.6%, p=0.75) were all similar. PSI was also similar globally (1507 vs 1402, p=0.34) as well as for CD4 (1577 vs 1509, p=0.78) and CD8 subsets (1437 vs 1248 p=0.31). In assessing contribution to PSI per cytokine, CD4 cells in the 8- vs 12-day arm had higher levels of perforin (116 vs 55, p=0.01). CD8 cells in the 8- vs 12-day arm had higher levels of MCP-1 (7.2 vs 1.2, p=0.02) and less IL-4 (13.3 vs 38.6, p=0.02).
Conclusions:
Bispecific LV20.19 CAR T-cells expanded in IL7+15 have greater polyfunctionality and polyfunctional strength indices than IL-2 expanded cells. Highly polyfunctional CAR T-cells and higher PSI have been associated with clinical response in single antigen targeted CAR T-cells (Rossi Blood 2018). Global polyfunctionality for LV20.19 CAR-T products with IL-7+15 was notably higher at 52% than previously reported polyfunctionality of 20-25% for single antigen targeted CAR T-cells. Manufacturing time (8- vs 12-days) did not have a significant impact on PFA or PSI. Expansion of LV20.19 CAR T-cells in IL7+15 may generate a more robust CAR-T product than expansion in IL-2, which could improve persistence, cytotoxicity, and ultimately patient outcomes. An ongoing prospective trial (NCT04186520) is evaluating clinical outcomes with IL-7+15 expanded LV20.19 CAR T-cells and is reported separately.
Fenske: Servier Pharmaceuticals: Consultancy; Bristol-Myers Squibb: Speakers Bureau; Pharmacyclics: Consultancy; Sanofi: Speakers Bureau; AbbVie: Consultancy; ADC Therapeutics: Consultancy; CSL Therapeutics: Consultancy; Seattle Genetics: Speakers Bureau; KaryoPharm: Consultancy; Adaptive Biotechnologies: Consultancy; AstraZeneca: Speakers Bureau; Kite (Gilead): Speakers Bureau; TG Therapeutics: Consultancy, Speakers Bureau; Beigene: Consultancy; Biogen: Consultancy; MorphoSys: Consultancy. Hamadani: Takeda, Spectrum Pharmaceuticals and Astellas Pharma: Research Funding; Sanofi, Genzyme, AstraZeneca, BeiGene: Speakers Bureau; Janssen, Incyte, ADC Therapeutics, Omeros, Morphosys, Kite: Consultancy. Schneider: Employee of Lentigen Technology, a Miltenyi Biotec Company: Current Employment. Hari: Karyopharm: Consultancy; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other, Research Funding, Speakers Bureau; Takeda: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other, Research Funding, Speakers Bureau; Sanofi: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other, Research Funding, Speakers Bureau; GSK: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other, Research Funding, Speakers Bureau; Adaptive Biotech: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Oncopeptides: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Millenium: Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene-BMS: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other, Research Funding, Speakers Bureau. Johnson: Miltenyi Biotec: Research Funding. Shah: Epizyme: Consultancy; Lily: Consultancy, Honoraria, Research Funding; Legend: Consultancy; Miltenyi Biotec: Consultancy, Honoraria, Research Funding; Kite: Consultancy; Umoja: Consultancy; Incyte: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal