Abstract
In acute myeloid leukemia (AML), relapse following standard chemotherapy is common, leading to 2-year survival rates of less than 30%. Relapse is caused by leukemia stem cells (LSCs), a rare population of mostly quiescent cells that are chemo-refractory and can recapitulate the disease. Our overall goal is to define mechanisms of self-renewal that could be targeted to cure AML and prevent relapse. Functionally, self-renewal is defined by the ability to propagate leukemia in vivo.
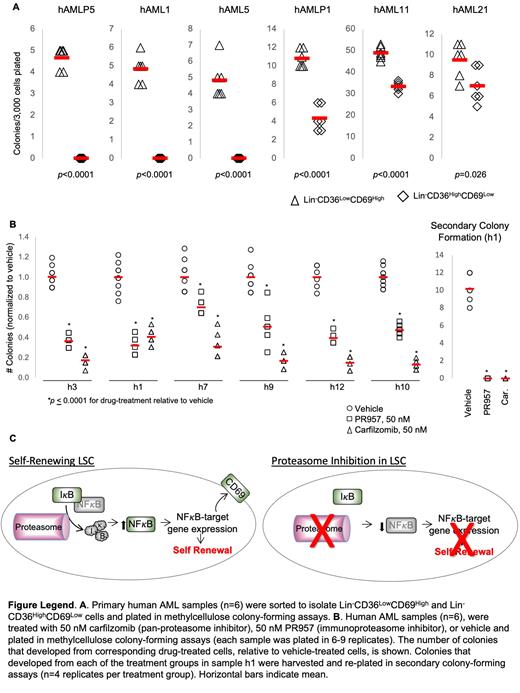
We use transcriptional and protein profiling to define the functional states of LSCs. We previously demonstrated that NRAS G12V facilitates self-renewal in a mouse model of AML (Mll-AF9/NRAS G12V). Using single-cell RNA sequencing and in vivo leukemia assays, we showed that the stem cell compartment in this model has two distinct subpopulations which differ in their self-renewal and proliferative abilities. The subset of LSCs marked by CD36 LowCD69 High (CD69 High) expression can self-renew and are poorly proliferative. The CD36 HighCD69 Low (CD36 High) subset is unable to self-renew and is highly proliferative. These data demonstrated that self-renewal and rapid proliferation are mutually exclusive functions among LSCs. We demonstrated that the gene expression profiles associated with these functionally distinct LSC subsets are also similarly differentially expressed in primary human LSCs, at the single-cell level (Sachs Cancer Research 2020). Given the functional differences between the CD36 LowCD69 High and CD36 HighCD69 Low subsets in murine AML, we tested whether CD69 and CD36 likewise discriminate self-renewal and proliferation in human AML. We sorted primary human AML samples according to CD69 and CD36 expression and found that Lin -CD69 High cells formed more colonies than Lin -CD36 High cells in every sample tested (n=6). Notably, in three of the six samples, the Lin -CD36 High population was unable to form any colonies (Panel A). These data suggest that CD69 may identify a self-renewing subset of human AML as it does in murine AML. Next, we asked whether Lin -CD69 High and Lin -CD36 High subsets harbor unique signaling protein activation profiles that could potentially be targeted therapeutically. We used CyTOF (mass cytometry) to compare the levels of 12 intracellular signaling proteins between these subpopulations in 14 human AML samples with intermediate and poor risk genetics. CyTOF quantitatively measures a panel of proteins at the single-cell level and allows us to profile activated signaling pathways within well-defined immunophenotypic subpopulations. We found upregulation of both total and phosphorylated (t- and p-) NF-kappa B, pERK, p4EBP1, pMAPKAPKII, and its upstream activator, pP38 in the Lin -CD69 High subset relative to the Lin -CD36 High. Notably, CD69 is a NF-kappa B target gene. Indeed, our single-cell transcriptional data showed that NF-kappa B target genes were upregulated in the self-renewing, CD69 High subset of murine and human LSCs (Sachs Cancer Research 2020). NF-kappa B is a well-known mediator of AML-self-renewal. Next, we asked whether targeting NF-kappa B with proteasome inhibitors might reduce self-renewal capacity in human AML. We plated six primary human samples in colony-forming assays with pan-proteasome inhibitor, carfilzomib, immunoproteasome inhibitor, PR957, or vehicle and found that both inhibitors reduced colony formation in every sample tested. We harvested colonies from each of the inhibitor and vehicle groups and plated them in secondary colony-forming assays. Colonies harvested from vehicle-treated samples formed colonies but those harvested from either carfilzomib or PR957-treated samples were unable to form any secondary colonies (Panel B). Using CyTOF, we validated that both inhibitors reduce NF-kappa B levels in these samples.
These data demonstrate that CD69 marks a self-renewing subset of human AML, as it does in our murine AML model, and suggest that the transcriptional mechanisms of self-renewal that we defined in our murine model at the single-cell level may be shared with human AML. Additionally, these data demonstrate enhanced NF-kappa B levels in this self-renewing subset and that targeting the NF-kappa B pathway with proteasome inhibitors attenuates self-renewal in primary human AML samples (Panel C). These findings suggest that proteasome inhibition may be an effective approach to treating and preventing relapse in AML.
No relevant conflicts of interest to declare.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal