Abstract
Background:
In the last decade there have been significant advances in diagnosing and classifying adult acute myeloid leukemia (AML) based on genomic profiling, enabling risk-stratification and targeted therapies. In 2017 the US FDA approved the first gene mutation targeted therapies for AML with multiple additional targeted therapies since approved or in development. Given the typical acuity of AML at initial presentation however and the current turnaround time for next-generation sequencing (NGS) assays, most patients will start definitive initial therapy before all potentially targetable mutations are known. There is, therefore, a significant need for a fast molecular genotyping test to determine eligibility for personalized therapy in AML.
The NCI Myeloid Assay (NMA) is a comprehensive targeted NGS assay on the Ion Torrent Genexus System, a fully automated platform that provides a rapid turnaround time from specimen receipt to clinical reporting. NMA utilizes Thermo Fisher Scientific's Oncomine Myeloid Assay GX and appears ideally suited for use in upcoming AML targeted therapy trials but has yet to be extensively tested in a cohort of AML patient diagnostic samples and compared to a standard targeted "myeloid panel" NGS assay platform (s-NGS).
Methods:
DNA samples (n=173) extracted from pretreatment bone marrow and/or peripheral blood of adult patients (n=112) diagnosed with de novo AML or high-risk myelodysplastic syndrome (MDS), were blindly tested in parallel using the NMA and s-NGS assays. For the NMA assay, 27.75ng of DNA was put into the Genexus System. All runs, controls, and samples were first analyzed for sequencing quality using established quality control (QC) metrics to assess pass/fail status. For all samples that passed QC metrics, variant results generated by the Ion Torrent Genexus pipeline were manually reviewed prior to being called true positive variants. For the s-NGS, using the ArcherDx Myeloid VariantPlex assay, a DNA input of 50ng was used for library preparation on a dual pre- and post-PCR separated automated liquid-handling workflow. Resulting libraries were sequencing on the Novaseq 6000 (Illumina) and the data analyzed using the Archer Analysis software and filtered as previously described (PMID: 34258102). Results from the two assays were compared for mutations with a variant allele fraction (VAF) >5% occurring in genes of interest in small molecule targeted clinical trials including: FLT3, IDH1, IDH2, JAK2, KIT, NPM1, NRAS, KRAS, and TP53. For FLT3-ITD comparison, the presence or absence of a call by the assay was used.
Results:
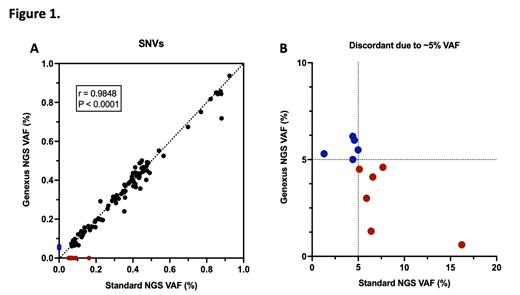
Utilizing a 5% VAF reporting threshold, a total of 171 and 174 variants were detected by NMA and s-NGS assays, respectively. A high rate of concordance was observed between the assays, with NMA detecting 96% of s-NGS variants and s-NGS detecting 95% of NMA variants. The VAF of detected single nucleotide variants was highly correlated (r=0.9848, P<0.0001, Figure 1A). NPM1 mutation VAF values trended lower by s-NGS compared to NMA.
We investigated the discordant calls (n=15 total in 11 patients). One patient was correctly identified as having an NRAS p.Gly12 mutation by both approaches, but the resulting mutation was incorrectly annotated by the s-NGS pipeline. Samples from two patients (including one with both blood and marrow tested) were correctly identified as being FLT3 tyrosine kinase domain mutated by both sequencing approaches, although only the major of two missense variants identified by s-NGS was reported by the NMA pipeline. None of these patients, however, would be misclassified. The remaining 11 discordant calls were false negatives (including 6 variants detected by s-NGS but not by NMA). All of these "edge case" variants were detectable by lowering the VAF reporting threshold below 5% (Figure 1B).
Conclusions:
NMA is an automated sample-to-results workflow that can identify myeloid disorder-associated genomic variants in less than 48 hours from library preparation to clinical reporting. We show that NMA is highly concordant with a standard DNA NGS assay for detecting mutations within recurrently mutated AML genes. Accurate rapid genotyping is required for assignment to initial treatment with targeted therapy, and this technology may be a valuable tool for upcoming clinical trials for patients with myeloid malignancies.
Zhang: Thermo Fisher Scientific: Current Employment. Sedova: Thermo Fisher Scientific: Current Employment. Huang: Thermo Fisher Scientific: Current Employment. Mittal: Thermo Fisher Scientific: Current Employment. Hatch: Thermo Fisher Scientific: Current Employment. Ni: Thermo Fisher Scientific: Current Employment. Kaznadzey: Thermo Fisher Scientific: Current Employment. Sadis: Thermo Fisher Scientific: Current Employment. Smith: Thermo Fisher Scientific: Current Employment. Williams: Illumina: Other: CRADA. Hourigan: Sellas: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal